Question
Problem 2. Match the structures of the following CHO isomers 1-5 with the IR spectra (A-E) shown below. For each problem, specify the key
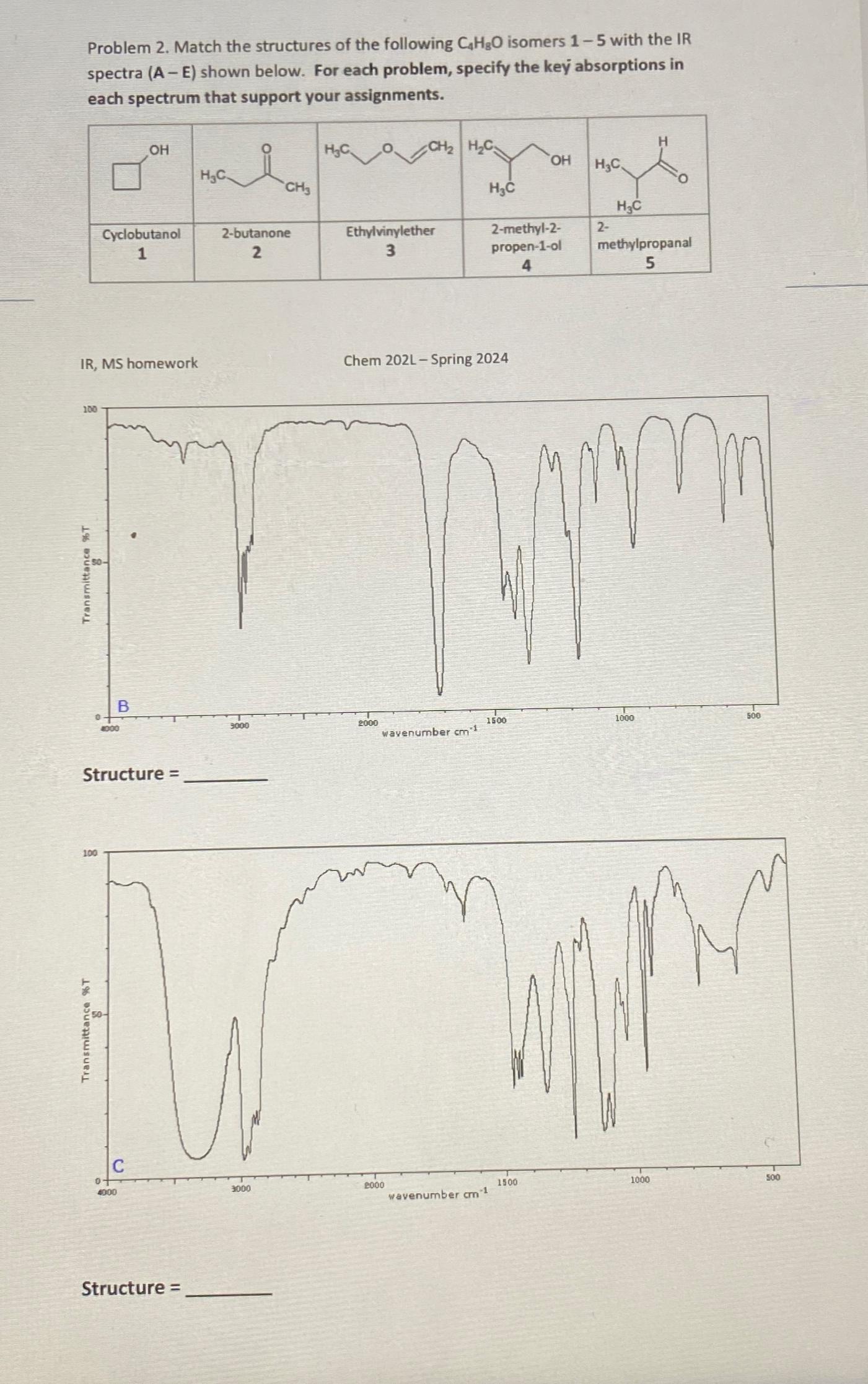
Problem 2. Match the structures of the following CHO isomers 1-5 with the IR spectra (A-E) shown below. For each problem, specify the key absorptions in each spectrum that support your assignments. 100 IR, MS homework Transmittance %T 0 100 Transmittance %T Cyclobutanol 1 4000 Structure = 0 OH B 4000 Structure = HC i 2-butanone 2 9000 CH3 3000 HC Ethylvinylether 3 CH HC 2000 Chem 202L - Spring 2024 wavenumber cm 1 2000 HC wavenumber cm-1 2-methyl-2- propen-1-ol 4 1500 OH 1500 HC 2- HC methylpropanal 5 mm 1000 1000 500 500
Step by Step Solution
There are 3 Steps involved in it
Step: 1
Solutions Step 1 The image shows two infraredIRspectra labeled B and C and we are asked to match the...
Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get StartedRecommended Textbook for
Organic Chemistry
Authors: Paula Yurkanis Bruice
4th edition
131407481, 978-0131407480
Students also viewed these Chemical Engineering questions
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
Question
Answered: 1 week ago
View Answer in SolutionInn App



