Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Thank you! Express your answer with the appropriate units. E= = 1.91x10-19 J Som Previous Answers Correct Correct answer is shown. Your answer 19.47-10-20 J
Thank you! 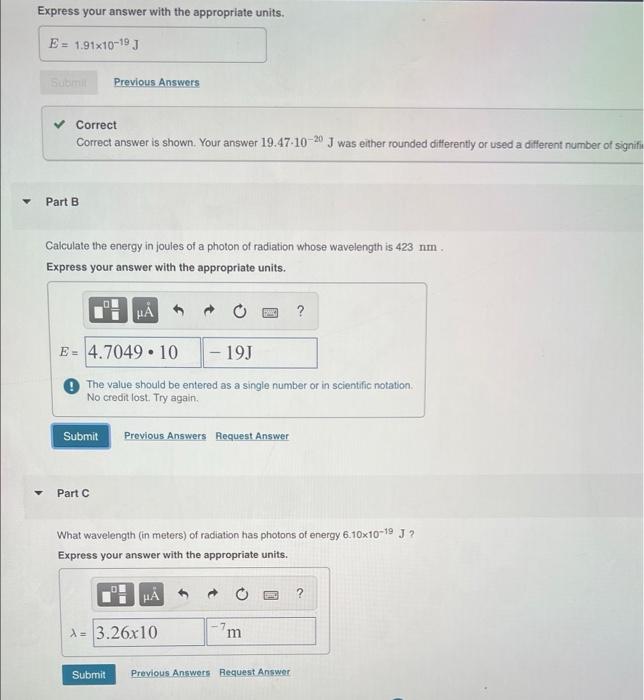
Express your answer with the appropriate units. E= = 1.91x10-19 J Som Previous Answers Correct Correct answer is shown. Your answer 19.47-10-20 J was either rounded differently or used a different number of signifi Part B Calculate the energy in joules of a photo of radiation whose wavelength is 423 nm Express your answer with the appropriate units. 6 PER ? E = 4.7049.10 - 19 The value should be entered as a single number or in scientific notation. No credit lost. Try again Submit Previous Answers Request Answer Part What wavelength (in meters) of radiation has photons of energy 6.10x10-19 J? Express your answer with the appropriate units. HA ? 1 = 3.26x10 - 7 m Submit Previous Answers Request 
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


