Answered step by step
Verified Expert Solution
Question
1 Approved Answer
The number of molecules of a monoatomic ideal gas is N and is constant. In all processes the compression work is quasi-static. The gas is
The number of molecules of a monoatomic ideal gas is N and is constant. In all processes the compression work is quasi-static. The gas is initially at temperature T and expands adiabatically from volume V to volume 2V. Show how much the mean square root velocity of the molecules (Vokh= ) has changed compared to the final state and the initial state, (Vokh_final/Vokh_first)=?
) has changed compared to the final state and the initial state, (Vokh_final/Vokh_first)=?
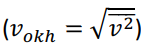
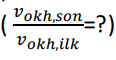 (vokh=v2) (vokh,ilkvokh,son=?) (vokh=v2) (vokh,ilkvokh,son=?)
(vokh=v2) (vokh,ilkvokh,son=?) (vokh=v2) (vokh,ilkvokh,son=?) Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


