Answered step by step
Verified Expert Solution
Question
1 Approved Answer
Time left 0:30:55 Consider the following apparatus. Calculate the partial pressures of helium and neon after the stopcock is open. The temperature remains constant at
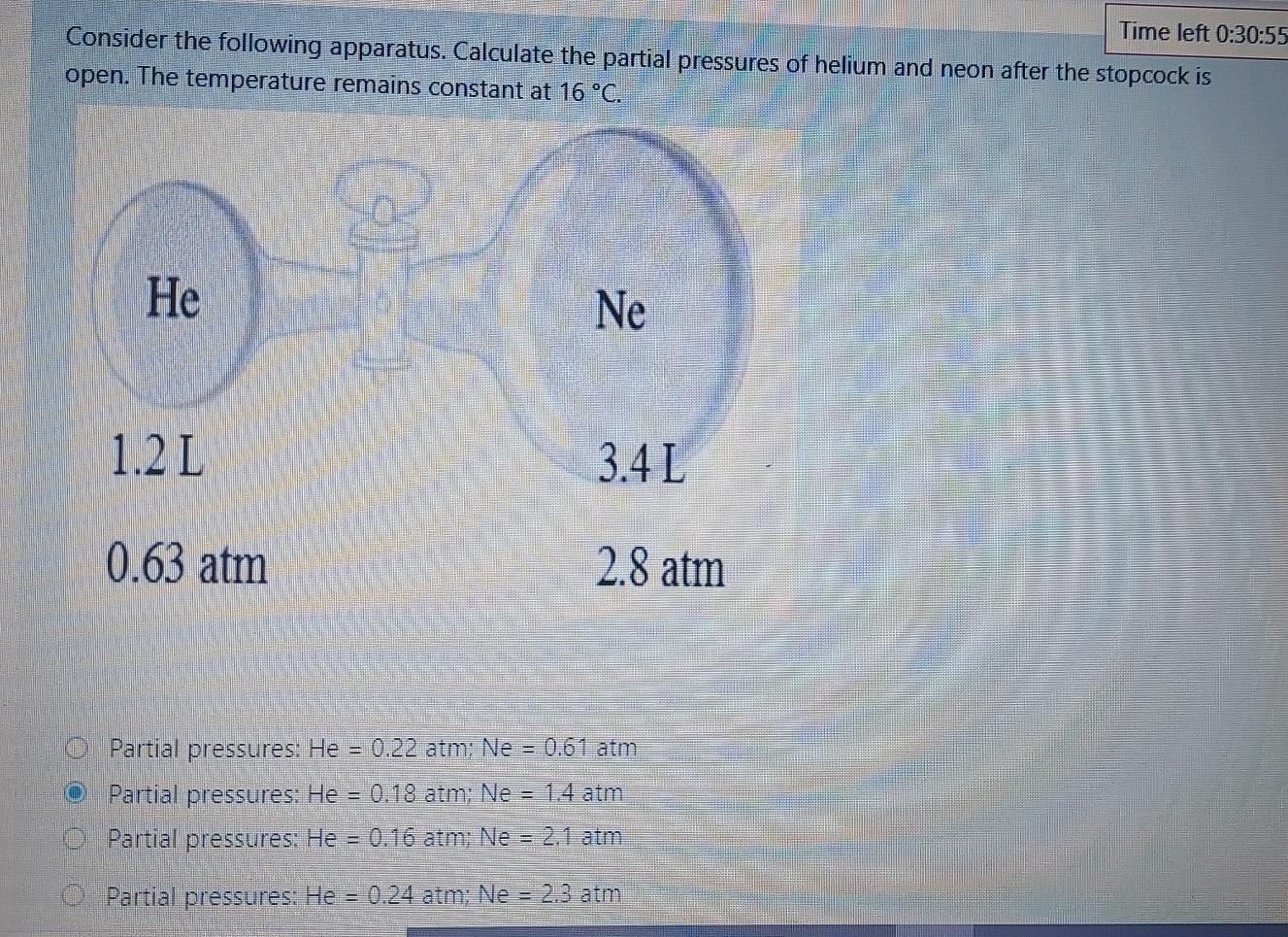
Time left 0:30:55 Consider the following apparatus. Calculate the partial pressures of helium and neon after the stopcock is open. The temperature remains constant at 16 C. He Ne 1.2 L 3.4 L 0.63 atm 2.8 atm Partial pressures: He = 0.22 atm; Ne = 0.61 atm Partial pressures: He = 0.18 atm; Ne = 1.4 atm 0 Partial pressures: He = 0.16 atm; Ne = 2.1 atm O Partial pressures: He 0.24 atm; Ne = 2.3 atm
Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


