Answered step by step
Verified Expert Solution
Question
1 Approved Answer
use matlab and screenshot the code this is x1_y1_P.txt 1. (25 pts) Activity coefficient y is an important thermodynamic parameter to describe nonideality of a
use matlab and screenshot the code 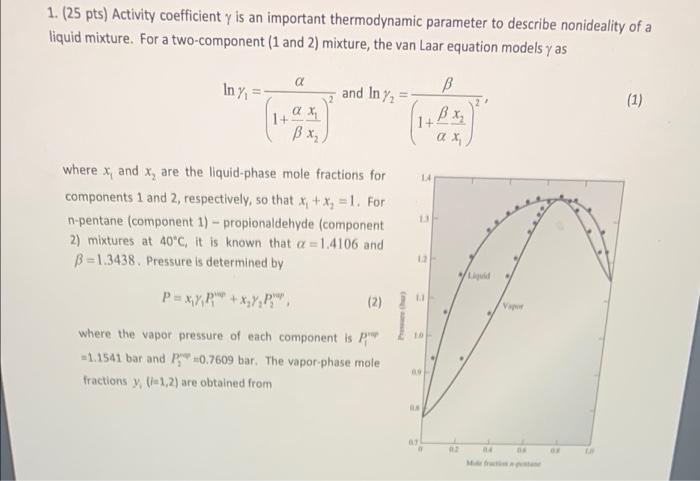
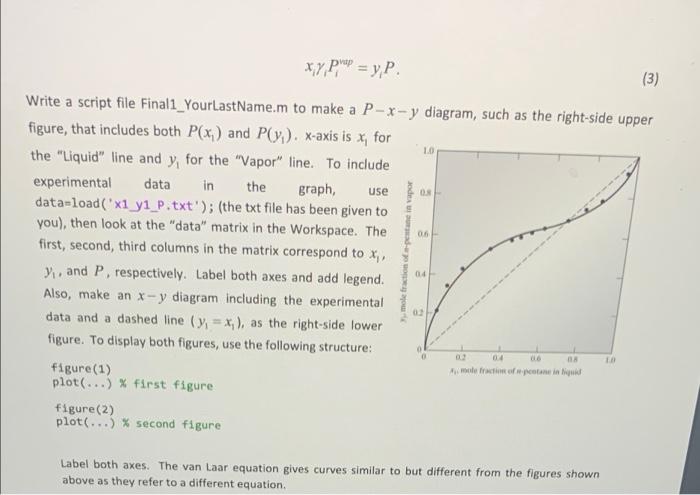

1. (25 pts) Activity coefficient y is an important thermodynamic parameter to describe nonideality of a liquid mixture. For a two-component (1 and 2) mixture, the van Laar equation models y as a In y and In Y B (1) , 1+ , ax 1.4 where x, and x, are the liquid-phase mole fractions for components 1 and 2, respectively, so that x1 + x3 =1. For n-pentane (component 1) - propionaldehyde (component 2) mixtures at 40C, it is known that a =1.4106 and B = 1.3438. Pressure is determined by 13 12 . P = + x,y, (2) V 10 where the vapor pressure of each component is p -1.1541 bar and p 0.7609 bar. The vapor phase mole fractions (+1,2) are obtained from xy P p = y, P. (3) LO use ode 0 06 Write a script file Finali_YourLastName.m to make a P-x-y diagram, such as the right-side upper figure, that includes both P(x) and Py). x-axis is x, for the "Liquid" line and y, for the "Vapor" line. To include experimental data in the graph, data-load("x1 yi_P.txt'); (the txt file has been given to you), then look at the "data" matrix in the Workspace. The first, second, third columns in the matrix correspond to x) y, and P, respectively. Label both axes and add legend. Also, make an x-y diagram including the experimental data and a dashed line (y) = x), as the right-side lower figure. To display both figures, use the following structure: figure (1) plot(...) % first figure figure(2) plot(...) % second figure 04 04 016 mole fraction of protein Label both axes. The van Laar equation gives curves similar to but different from the figures shown above as they refer to a different equation, 00 0.7609 0.0503 0.2121 0.9398 0.1014 0.3452 1.0643 0.1647 0.4288 1.1622 0.2212 0.4685 1.2173 0.3019 0.5281 1.2756 0.3476 0.5539 1.2949 0.4082 0.5686 1.3197 0.4463 0.5877 1.3354 0.5031 0.6146 1.3494 0.561 0.6311 1.3568 0.6812 0.6827 1.3636 0.7597 0.7293 1.3567 0.8333 0.7669 1.3353 0.918 0.8452 1.2814 1 1 1.1541 

this is x1_y1_P.txt

Step by Step Solution
There are 3 Steps involved in it
Step: 1

Get Instant Access to Expert-Tailored Solutions
See step-by-step solutions with expert insights and AI powered tools for academic success
Step: 2

Step: 3

Ace Your Homework with AI
Get the answers you need in no time with our AI-driven, step-by-step assistance
Get Started


