Given the density of 2.655 g/cm 3 of an SiO 2 glass, estimate Va in 3
Question:
Given the density of 2.655 g/cm3 of an SiO2 glass, estimate Va in Å3 per formula unit. Use the refractive index of 1.547 to calculate the polarization with the Lorentz–Lorenz equation (15.4).
Equation (15.4)
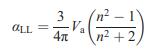
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:





