Give the expected product(s) of each of the following reactions. 1. LDA, THF 2. BICH,COCH, H 1.
Question:
Give the expected product(s) of each of the following reactions.
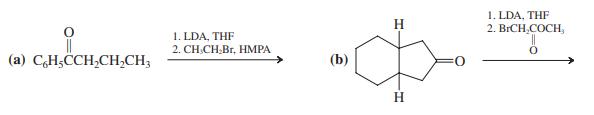
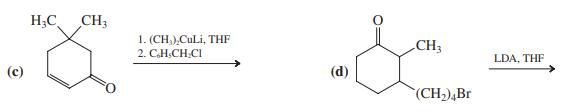
Transcribed Image Text:
1. LDA, THF 2. BICH,COCH, H 1. LDA, THF 2. CH.CH Br, HMPA (a) CH,CCH,CH,CH3 (b) 0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
LDA is lithium diisopropyl amide strong base which abstracts the ...View the full answer

Answered By

Prajoy Kumar Mitra
Well being honest I want to say that Chemistry is my first priority. I can help you in every possible way in the field of Chemistry. I believe knowledge needs to be shared. And regarding experience I would like to say that, recently I have completed my Masters in Chemistry & also Qualified the GATE exam. Though I don't have the teaching experience but I do have good teaching skills along with sufficient knowledge. And I'm confident enough to give you a better concept in Chemistry along with certain entrance preparations.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Write the expected product(s) of each of the following reactions. CH3 3 NANH, liquid NH, 2 NANH,, liquid NH, () CH-CH-CHCCH-CI (b) CH,OCH,CH,CH,CHCHCH3 I Br Br NaOCH, (1 equivalent), CH,OH CH3 CI CI...
-
Give the major product(s) of each of the following reactions. (a) (b) (c) (d) (e) (f) H;C I HO H
-
Give the product(s) of each of the following reactions, ignoring stereoisomers: (a) (b) (c) (d) (e) (f) hv CH CH,C CHCHNBS peroxide CH h CH3CH-CHCH.CH-CH3 Br2 + hv + Cl CH2Cl2 +Cl2 CH3 + Cl2
-
Presented below is information related to Larkspur Inc.'s inventory, assuming Larkspur uses lower-of-LIFO cost-or-market. (per unit) Skis Boots Parkas Historical cost $224.20 $125.08 $62.54 Selling...
-
Summarize the factual dispute in Lorilland Tobacco Co. v. Reilly. The case syllabus (summary) is found in Appendix VII
-
For what kinds of problems will modeling lead to an ODE? To a PDE?
-
1 What types of IS do different people in the organisation use perhaps locating them on the grid provided in Figure 12.6?
-
A diesel-powered tractor with a cost of $214,000 and estimated residual value of $30,000 is expected to have a useful operating life of 50,000 hours. During January, the tractor was operated 175...
-
a company manufactures plastic products direct labor costs
-
A histogram of the n = 345 grade point averages reported by students in the Student Survey dataset is shown in Figure 2.28. (a) Estimate and interpret the 10th percentile and the 75th percentile. (b)...
-
Write the expected major product of reaction of each of the carbonyl compounds (i) (iii) with each of the reagents (a) (h). CH2CH,CH3 (i) (ii) CH;CH=C. (iii) CH || () , P, ,C- (c) Cl2, CCl, (e)...
-
Write the products of each of the following reactions after aqueous work-up. (e) Write the results that you expect from base treatment of the products of reactions (c) and (d). LDA, THE (a) CH;CCH; +...
-
Write the chemical formulas for the following compounds: (a) Aluminum hydroxide (b) Potassium sulfate (c) Copper(I) oxide (d) Zinc nitrate (e) Mercury(II) bromide (f) Iron(III) carbonate (g) Sodium...
-
Add F1 and F2 using graphical method, (triangle or parallelogram) Determine: 1 Magnitude,2. Direction measured CCW from positive axis, im now to America need help. CoursHeroTranscribedText 20 F-SON...
-
What is Monetary Policy? What is Monetary Base or High Powered Money? How commercial Banks create money Supply? Hint: By giving loans through creating checking account What is deposit multiplier?...
-
2 4 . In the current year, Madison sold Section 1 2 4 5 property for $ 6 , 0 0 0 . The property cost $ 2 6 , 0 0 0 when it was purchased 5 years ago. The depreciation claimed on the property was $ 2...
-
Swifty Company purchased machinery on January 1, 2025, for $82,400. The machinery is estimated to have a salvage value of $8,240 after a useful life of 8 years. (a) Your answer is incorrect. Compute...
-
Currently, the unit selling price is $ 5 0 , the variable cost is $ 3 4 , and the total fixed costs are $ 1 0 8 , 0 0 0 . a . Compute the current break - even sales in units.
-
Obtain the Target Corporations annual report for its 2018 fiscal year (year ended February 2, 2019) at http://investors.target.com using the instructions in Appendix A, and use it to answer the...
-
Cable Corporation is 60% owned by Anna and 40% owned by Jim, who are unrelated. It has noncash assets, which it sells to an unrelated purchaser for $100,000 in cash and $900,000 in installment...
-
A monolayer of CO molecules (effective area 0.165 nm3) is adsorbed on the surface of 1.00 g of an Fe/Al203 catalyst at 77 K, the boiling point of liquid nitrogen. Upon warming, the carbon monoxide...
-
The volume of gas at 20C and 1.00 bar adsorbed on the surface of 1.50 g of a sample of silica at On C was 1.60 cm-3 at 52.4 kPa and 2.73 cm3 at 104kPa. What is the value of Vmon?
-
The enthalpy of adsorption of ammonia on a nickel surface is found to be -155 k] mol-1 Estimate the mean lifetime of an NH3 molecule on the surface at 500 K.
-
Y de cunto esperas que fuera esta disminucin? Cunto bajara el precio de la accin despus de la fecha de ex-dividendo? (Ingresa tu respuesta con dos decimales.)
-
A machine that can be purchased for cash today for 100,000, is leased for 3 years, for a down payment of 30,000, plus 3 annual end of year payments of 30,000 each. If the residual value is 25,000,...
-
A food service operation has an average meal price of $8.00. Variable costs are $4.25 per meal and fixed costs are $10,000. How many meals must be sold to provide a pre-tax income of $20,000?

Study smarter with the SolutionInn App


