How would each reaction in Problem 25 be affected by the addition of each of the following
Question:
How would each reaction in Problem 25 be affected by the addition of each of the following substances to the solvolysis mixture?
(a) H2O
(b) KI
(c) NaN3
(d) CH3CH2OCH2CH3
In Problem 25
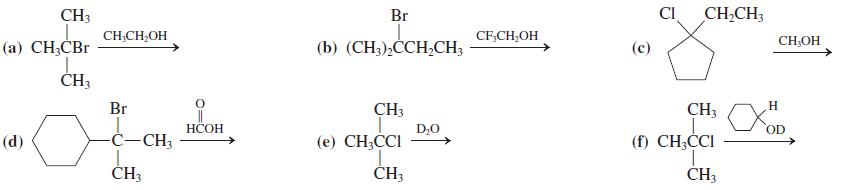
Transcribed Image Text:
CH3 Br CI CH,CH; CH;CH,OH CF,CH;OH CH;OH (а) CН,СВr (b) (CH).ССH-CH; (с) ČH3 Br CH3 CH3 H НСОН D;0 OD (d) -Ċ-CH3 (e) CH;CCI (f) CH;CI CH3 ČH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
a This would result in a mixture of ether ...View the full answer

Answered By

Akop Yepremyan
My aim would be to adequately walk through a given problem and find a solution through logic, rather than memorizing each question. With my strong background in tutoring and research as a PhD chemist from Texas Christian University (TCU) I will provide both theoretical and practical knowledge for your success.
I have been studying, teaching and researching in the field of chemistry for over 15 years. I have mentored and conducted multiple review sessions for students. Because of my extensive laboratory experience, I allow the student the visualize a given problem, thus making them much more enjoyable and easier to solve.
I am happy to teach you the fundamentals of chemistry in a way that will be very enjoyable for you and you are not counting the minutes to stop studying. Chemistry takes time to learn, I will make sure you push through the barrier that makes chemistry a difficult subject, and reach a level where you are very comfortable with solving any problem that is presented to you. I am available to help anytime and will respond to your need promptly.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
How would the solubility of calcium fluoride be affected by the presence of fluoride ion from another source? What is the solubility of calcium fluoride in a saturated solution of barium fluoride?...
-
The following reaction is spontaneous in the direction given. A(g) + B(g) C(g) + D(g) Suppose you are given a vessel containing an equilibrium mixture of A, B, C, and D, and you increase the...
-
Consider the binding reaction L + R LR, where L is a ligand and R is its receptor. When 1 X 10 -3 M L is added to a solution containing 5 X 10 -2 M R, 90 percent of the L binds to form LR. What is...
-
Given the following network, with the indicated flow capacities along each branch, determine the maximum flow from source node 1 to destination node 10 and the flow along eachpath: 15 5 10 7 16 12 0...
-
What is the quality audit used for?
-
2. Test whether there is an association between a person's educational attainment and how much television they watch. Use the GSS08 data set to perform an ANOVA on respondents' highest educational...
-
Modifying default configurations to improve security is called a. encryption hardening b. patching dialing c. hardening d. war dialing
-
The stockholders equity accounts of Castle Corporation on January 1, 2014, were as follows. Preferred Stock (8%, $50 par, cumulative, 10,000 shares authorized) ......$ 400,000 Common Stock ($1 stated...
-
Increasing the sophistication and complexity of an automated system increase the chance of system failures because? A. programs that run complex automation systems are not readily observable by the...
-
case 1 Swift Logistics, an international supply chain group, has been operating in Hong Kong for a long time. It possesses a large network and serves diverse customers from various Asian countries...
-
Classify each of the following common objects as being either chiral or achiral. Assume in each case that the object is in its simplest form, without decoration or printed labels. (a) A ladder; (b) A...
-
Give the major product(s) of the following reactions. Indicate which of the following mechanism(s) is in operation: S N 1, S N 2, E1, or E2. If no reaction takes place, write no reaction.
-
The spreadsheet provides data about the demand for money in Minland. Columns A and B show the demand for money schedule when real GDP ( Y0 ) is $10 billion and Columns A and C show the demand for...
-
What is the relationship between UCs and Modes of Operations?
-
What are the relationships among Phases, Modes, and States of Operation?
-
How do we derive Modes and States?
-
When are Modes and States defined in a project? Why?
-
Do Modes contain States or do States contain Modes?
-
A structural component in the form of a wide plate is to be fabricated from a steel alloy that has a plane-strain fracture toughness of 98.9 MPa(m (90 ksi(in) and a yield strength of 860 MPa (125,000...
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
The use of supercritical fluids as mobile phases in SFC depends on their properties as non polar solvents. The solubility parameter, , is defined as (Ucohesive/Vm)1/2, where Ucohesive is the cohesive...
-
Calculate the maximum non-expansion work per mole that may be obtained from a fuel cell in which the chemical reaction is the combustion of propane at 298 K.
-
A certain heat engine operates between 1000 K and 500 K. (a) What is the maximum efficiency of the engine? (b) Calculate the maximum work that can be done by for each 1.0 k] of heat supplied by the...
-
A random sample contains 28 observations. Determine the p-VALUE for the value of the calculated test statistic below: Null: mu = 50 Alternative: mu not = 50 e. t = -2.2911 Correct answer is 0.02 <...
-
The direct delivery of goods to stores and to your home and in today's climate of home delivery, the options for answering these questions are everywhere. 1. What kind of retailers would be best...
-
3. Let's practice time-series forecasting of new home sales. Click here (https://www.census.gov/construction/nrs/historical_data/index.html) to see the newest data in the first table: Houses Sold...

Study smarter with the SolutionInn App


