o-Iodoaniline is the common name of which of the following compounds? NH2 CH3 NH2 NH2 (a) (b)
Question:
o-Iodoaniline is the common name of which of the following compounds?
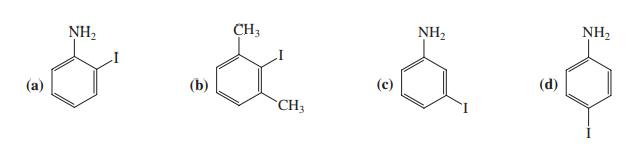
Transcribed Image Text:
NH2 CH3 NH2 NH2 (a) (b) (d) CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
oIodoaniline is the common name of the ...View the full answer

Answered By

Aditi Deswal
Currently , I am doing post graduation ( MSc.) in Chemistry . I want to bacome a lecturer . I feel happy when I teach student and solve their problem . It is my passion as well as hobby . I feel blessed If I got a chance to share my knowledge as much as I have . I teach student at my home also. I love teching and want to spend my whole life to teach the students and explores new things.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
o-Iodoaniline is the common name of which of the following compounds? NH2 CH3 NH2 NH2 (a) (b) (d) CH3
-
Cubane (C8H8) is the common name of a polycyclic hydrocarbon that was first synthesized in the early 1960s. As its name implies, its structure is that of a cube. How many rings are present in cubane?...
-
Cubane (C8H8) is the common name of a polycyclic hydrocarbon that was first synthesized in the early 1960s. As its name implies, its structure is that of a cube. How many rings are present in Cubane?
-
Suppose that the weight of a typical American male follows a normal distribution with = 180 lb and = 30 lb. Also, suppose 91.92% of all American males weigh more than I weigh. a. What fraction of...
-
Which of the three organizational structures are organizations gravitating toward in order to improve their project success rates? Why?
-
Find the temperature distribution T(x, y) and the complex potential F(z) in the given thin metal plate whose faces are insulated and whose edges are kept at the indicated temperatures or are...
-
Outline the major methods available to improve emotional welfare among staff? LO1
-
Grahams Steel Parts produces parts for the automobile industry. The company has monthly fixed costs of $630,000 and a contribution margin of 80% of revenues. Requirements 1. Compute Grahams monthly...
-
Compute the discounted payback statistic for Project C if the appropriate cost of capital is 6 percent and the maximum allowable discounted payback period is three years. (Do not round intermediate...
-
Bonds 1. Municipal Bonds - Municipal bonds are haircut per Exhibit 1 based on both their time to maturity and scheduled maturity at date of issue. 2. Corporate Bonds - Corporate bonds are haircut...
-
As a team, discuss the following complementary experimental results as they pertain to the mechanism of electrophilic aromatic substitution. (a) A solution of HCl and benzene is colorless and does...
-
The species that is not aromatic according to Hckels rule is () (b) () (d)
-
N + 2 equal masses hang from a system of pulleys, as shown in Fig. What are the accelerations of all themasses? N=3
-
If the 230-lb block is released from rest when the spring is unstretched, determine the velocity of the block after it has descended 5ft . The drum has a weight of 70lb and a radius of gyration of...
-
A B C D E F 1 Frequency : Monthly 2 Loan Amount: 150000.00 3 Interest Rate: 7.25% 4 Term(years): 30.00 5 No. of payments in a year: 12 time(s) 6 Periodic Rate: 0.60% =C3/C5 7 Total number of payment:...
-
A company which manufactures microwaves advertises that 90% of their microwaves are flawless, requiring no adjustments. Their quality control department tests this percentage on a regular basis. On...
-
A new retail store is being planned for a site that contains 40 ft of soft clay (c 0.075 ft2/day, y = 100 pcf). The clay layer is overlain by 15 ft of sand (y = 112 pcf) and is underlain by dense...
-
Perez Bags (PB) is a designer of high-quality backpacks and purses. Each design is made in small batches. Each spring, PB comes out with new designs for the backpack and for the purse. The company...
-
Compute the specified ratios using Duluth Companys balance sheet for Year 3. Assets Cash .................................................................................... $ 36,000 Marketable...
-
In a paragraph of approximately 150-200 words, analyze a film or TV/Streaming Show poster of your choosing by focusing on the ways in which representations in the poster are gendered. Include an...
-
The scattering of electrons or neutrons from a pair of nuclei separated by a distance Rij and orientated at a definite angle to the incident beam can be calculated. When the molecule consists of a...
-
D. Sellmann, M.W. Wemple, W. Dona Bauer, and F.W. Heinemann (Inorg. Chem. 36, 1397 (1997)) describe the synthesis and reactivity of the ruthenium nitride compound [N(C4H9)4] [Ru(N)(S2C6H4)2J. The...
-
In an intrinsic semiconductor, the band gap is so small that the Fermi-Dirac distribution results in some electrons populating the conduction band. It follows from the exponential form of the...
-
Oscar Corporation generated $1 million of taxable income. Oscars activities and sales are restricted to State P, which imposes a 10% income tax. 100% of the stock of Oscar is owned by Felix...
-
Attempts: Keep the Highest: 12 6. Checking account reconciliation How do you balance your checkbook? Your roommate, Felix, has never before had a checking account, but he finally opened one last...
-
Spanish Peaks Railroad Inc. is considering acquiring equipment at a cost of $124,000. The equipment has an estimated life of 10 years and no residual value. It is expected to provide yearly net cash...

Study smarter with the SolutionInn App


