The reaction of ethyl butanoate with sodium ethoxide in CH 3 CH 2 OH gives (a) (b)
Question:
The reaction of ethyl butanoate with sodium ethoxide in CH3CH2OH gives
(a)
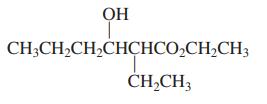
(b)
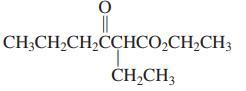
(c)
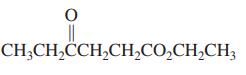
(d)
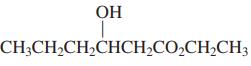
Transcribed Image Text:
ОН CH;CH,CH,CHCHCO,CH,CH3 ČH,CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Base catalysed condensation of same ester containing alfa hydrogen atom is ...View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Compound A (C7H13Br) is a tertiary bromide. On treatment with sodium ethoxide in ethanol, A is converted into B (C7H12). Ozonolysis of B gives C as the only product. Deduce the structures of A and B....
-
Give the mechanistic symbols (SN1, SN2, E1, E2) that are most consistent with each of the following statements: (a) Methyl halides react with sodium ethoxide in ethanol only by this mechanism. (b)...
-
Give the structure of the expected product from the reaction of isopropyl benzene with (a) Hydrogen (3 mol), Pt (b) Sodium and ethanol in liquid ammonia (c) Sodium dichromate, water, sulfuric acid,...
-
If you sign up for a savings account with an annual interest rate of 12% (1% per month) that you will receive 4 million won in 2 years, what is the monthly amount you have to pay? However, suppose...
-
Consider the two economies shown in Figure 3.1. When there is free trade, are we sure that each country should specialize completely in producing only one of the products? For instance, perhaps each...
-
Solve each problem. |x-1| = |2x + 3|
-
Herberger Oil & Gas Company paid \($10\) million for the drilling rights to a 1,000 acre tract of land near Midland, Texas. On the basis of several exploratory wells that had cost an aggregate of...
-
Master Delivery Inc. is considering the purchase of an additional delivery truck for $90,000 on January 1, 20Y4. The truck is expected to have a five-year life with an expected residual value of...
-
1. Using the General Model of the Operation and the steps therein as your framework, describe how your organisation delivers goods and/or services to the end consumer. Use appropriate examples to...
-
Agrico Inc. accepted a 10-month, 12.8% (annual rate), $9,000 note from one of its customers on May 15, 2019; interest is payable with the principal at maturity. Required: a. Use the horizontal model...
-
Two of the following four compounds are more acidic than CH 3 OH (i.e., two of these have K a greater than methanol). Which ones? (a) A and B; (b) B and C; (c) C and D; (d) D and A; (e) D and B....
-
When acid A (margin) is heated to 230C, CO 2 and H 2 O are evolved and a new compound is formed. Which one? (a) (b) HO 2 CCH 2 CH 2 CH 2 CH 3 (c) (d) CH 3 CH 2 CH(CO 2 H) 2 (e) CO,H HO,C(CH2),CH CO,H
-
What is the distinction between a port set in Mach and a port group in CHORUS?
-
How does the auditors assessment of business risk affect (1) the assignment of personnel to an engagement and (2) the auditors responsibility for conducting the audit to search for material fraud?
-
How does a financial defalcation differ from fraudulent financial reporting? Explain. Do the auditors responsibilities differ for detecting these two types of client fraud?
-
Describe how the objectives approach to evaluating the adequacy of controls in an accounting application is implemented.
-
Residual audit risk is most closely related to the concept of: a. Control risk b. Audit risk c. Detection risk d. Inherent risk
-
What is the difference between a standard external audit report on internal control and the newly designed SysTrust report on computer controls? Why is this difference important?
-
Remember to insert units when writing your answers. For following Exercises, complete the chart. 1. 2. Tons Pounds Object Ounces Statue of Liberty-weight of copper sheeting 100 Statue of...
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
Assign formal charger to the atoms in each of the followingmolecules: ( NNEN: CH (c) HCN3DN-DN: (a) H3C-N-O: CH
-
Maleic acid has a dipole moment, but the closely related fumaric acid, a substance involved in the citric acid cycle by which food molecules are metabolized, does not.Explain - C-OH - C=C c=C ...
-
Rank the following substances in order of increasing acidity: C CH3CH3 CHCCH2CCH Acetic acid (pKa = 4.76) Phenol Acetone (pKa = 19.3) 2,4-Pentanedione (pKa = 9) (pKa = 9.9) %3D %3D
-
Hi this is a question about business finance, thank you!
-
You interviewed a new client and these were the facts provided. Provide them with a brief explanation of tax reason for each Their home mortgage interest is $ 1 2 0 0 , real estate taxes $ 8 , 2 0 0...
-
Part A Capital Budgeting Background: Electro Motors (Electro) is considering a new project to produce electric vehicles for the Australian domestic market and international markets. It has identified...

Study smarter with the SolutionInn App


