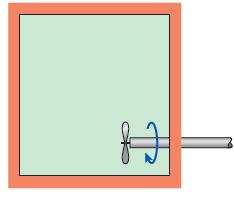
A substance is contained in a well-insulated rigid container that is equipped with a stirring device, as
Question:
A substance is contained in a well-insulated rigid container that is equipped with a stirring device, as shown in Fig. P4–30. Determine the change in the internal energy of this substance when 15 kJ of work is applied to the stirring device.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
A substance is contained in a wellinsulated rigid container tha...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
A 15 L tank contains 1 kg of R-12 refrigerant at 100oC. It is heated until the temperature of the refrigerant reaches 150oC. Determine the change in. (a) Internal energy (U). (b) Entropy, (S). Use...
-
A rigid container equipped with a stirring device contains 2.5 kg of motor oil. Determine the rate of specific energy increase when heat is transferred to the oil at a rate of 1 W, and 1.5 W of power...
-
In a refrigerator, 2.00 mol of an ideal monatomic gas are taken through the cycle shown in the figure. The temperature at point A is 800.0 K. (a) What are the temperature and pressure at point D? (b)...
-
Discuss why marketing management is important to an organization, and how corporate and business strategy analysis gives an organization a competitive advantage.
-
The owner of a restaurant serving Continental-style entrees has the business objective of learning more about the patterns of patron demand during the Friday-to-Sunday weekend time period. Data were...
-
Public library. Chicago Data Portal stores data for materials pulled to fulfill patron holds. Chicago Public Library is in 81 locations, including over 70 neighborhood branches and Sulzer, Legler,...
-
Deceiving subjects. Students sign up to be subjects in a psychology experiment. When they arrive, they are told that interviews are running late and are taken to a waiting room. The experimenters...
-
Describe the relationship between relevance and accuracy.
-
The CVP income statements shown below are available for Billings Company and Bozeman Company. Billings Co. Bozeman Co. $ 848,000 $ 848,000 Sales revenue Variable costs Contribution margin 354,000...
-
A rubber cylinder R of length L and cross-sectional area A is compressed inside a steel cylinder by a force F that applies a uniformly distributed pressure to the rubber (see figure). (a) Derive a...
-
Complete each line of the following table on the basis of the conservation of energy principle for a closed system. W. E, E, e2- e; kJ/kg m out kJ kJ kJ kJ kg 280 1020 550 860 3 5 -350 130 260 -150...
-
A rigid 1-ft 3 vessel contains R-134a originally at 20F and 27.7 percent quality. The refrigerant is then heated until its temperature is 100F. Calculate the heat transfer required to do this. Heat...
-
How might budgetary control be exercised over the procurement business process? Give specific examples.
-
The four classic leadership styles There are four leadership styles which are prominent in today's businesses and companies. They are Laissez-faire, Autocratic, Democratic, and Charismatic...
-
How can conflict be viewed positively? Cite a specific example of when this might be the case. Under what circumstances might "avoiding" conflict be "managing" conflict? In other words, when might...
-
Visit the website and answer the questions below. https://www.forbes.com/advisor/business/software/best-crm-small-business/ Based on the CRM software discussed in the article, which 3 software...
-
Planning consists of translating and organizations mission and vision into objectives. The organization's purpose is expressed as a mission statement, and what it becomes is expressed as a vision...
-
Question 1- Visit the Boots and Hearts Festival website: www.bootsandhearts.com. Using the information you find on the site, make an analysis of the festival's Strengths, Weaknesses, Opportunities...
-
If p 2 q = 4p - 2, find dp/dq.
-
Time Solutions, Inc. is an employment services firm that places both temporary and permanent workers with a variety of clients. Temporary placements account for 70% of Time Solutions' revenue;...
-
Saturated steam coming off the turbine of a steam power plant at 40oC condenses on the outside of a 3-cmouter- diameter, 35-m-long tube at a rate of 130 kg/h. Determine the rate of heat transfer from...
-
Water in a 5 - cm-deep pan is observed to boil at 98oC. At what temperature will the water in a 40 - cm-deep pan boil? Assume both pans are full of water.
-
A cooking pan whose inner diameter is 20 cm is filled with water and covered with a 4-kg lid. If the local atmospheric pressure is 101 kPa, determine the temperature at which the water starts boiling...
-
3. The nominal interest rate compounded monthly when your $7,000 becomes $11,700 in eight years is ________
-
An investor can design a risky portfolio based on two stocks, A and B. Stock A has an expected return of 21% and a standard deviation of return of 39%. Stock B has an expected return of 14% and a...
-
Advanced Small Business Certifica Drag and Drop the highlighted items into the correct boxes depending on whether they increase or decrease Alex's stock basis. Note your answers- you'll need them for...

Study smarter with the SolutionInn App


