A rigid 1-ft 3 vessel contains R-134a originally at 20F and 27.7 percent quality. The refrigerant is
Question:
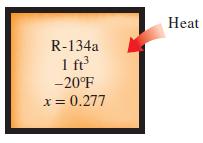
A rigid 1-ft3 vessel contains R-134a originally at –20°F and 27.7 percent quality. The refrigerant is then heated until its temperature is 100°F. Calculate the heat transfer required to do this.
Transcribed Image Text:
Heat R-134a 1 fr -20°F x= 0.277
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
R134a contained in a rigid vessel is heated The heat transfer is to be determined ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
A rigid 10-L vessel initially contains a mixture of liquid water and vapor at 100oC with 12.3 percent quality. The mixture is then heated until its temperature is 150oC. Calculate the heat transfer...
-
Calculate the heat transfer for the process described. Two springs with same spring constant are installed in a mass less piston/cylinder with the outside air at 100 kPa. If the piston is at the...
-
A piston/cylinder arrangement has the piston loaded with outside atmospheric pressure and the piston mass to a pressure of 20 lbf/in2. It contains water at 25 F, which is then heated until the water...
-
1. Resolve Class C 192.168.23.36 /27 2. Design DMZ minimumof 4 servers assigning an IP addressfor all devices 3. List and explain 4 primary servers that will be in the DMZ 4. Resolve Class C 192.168...
-
When performing a x2 test of independence in a contingency table with r rows and c columns, determine the upper-tail critical value of the test statistic in each of the following circumstances: a. a...
-
The trait model of leadership, although often discredited, continues to play a part in our understanding of leadership. LO1
-
Tempting subjects. A psychologist conducts the following experiment: she measures the attitude of subjects toward cheating, then has them play a game rigged so that winning without cheating is...
-
You are considering the choice between investing $50,000 in a conventional 1-year bank CD offering an interest rate of 5% and a 1-year Inflation-Plus CD offering 1.5% per year plus the rate of...
-
lew Policies Eurrent Attempt in Progress Carla Vista Corporation issued $350,000,9%, 20-year bonds on January 1, 2020, for $320,202. This price resulted in an effective interest rate of 10% on the...
-
Aristocrat , Baker , and Chef have formed Chez Guevara, Inc . ( Chez ) as a C corporation to operate a gourmet restaurant and bakery previously operated by Chef as a sole proprietorship. Aristocrat...
-
A substance is contained in a well-insulated rigid container that is equipped with a stirring device, as shown in Fig. P430. Determine the change in the internal energy of this substance when 15 kJ...
-
2 kg of saturated liquid water at 150C is heated at constant pressure in a pistoncylinder device until it is saturated vapor. Determine the heat transfer required for this process.
-
"In the future, I envision more companies moving toward actual cost systems. Standard cost variance analysis will no longer be required because inventory transactions will be recorded at actual cost....
-
You will be creating a Performance Improvement Plan to address an employee in the attached case study (see below). This is a scenario you may encounter in your future HR profession, so this...
-
For this prompt, consider your academic goals, including (but not limited to) such topics as how you plan to manage your time to fit in your studies; how you will build your skills, as needed; how...
-
1. An introduction of you as a leader (whether or not you see yourself as a leader, whether or not you like being a leader, what kinds of leadership roles you have had, etc.). 2. Summarize your...
-
Briefly, describe the firm in terms of the following items. a. Size in terms of market capitalization, annual revenue, number of employees, location(s). b. Discuss the financial position of the firm....
-
HealthyLife (HL) is a publicly-traded company in the Food Manufacturing Industry. HealthyLife has been around since the 1970s, and is mainly focused on the production and wholesale of "organic and...
-
Find the derivatives of the functions in Problem. y = 2ex
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
Reconsider Prob. 3-46. Using EES (or other) software, investigate the effect of the mass of the lid on the boiling temperature of water in the pan. Let the mass vary from 1 kg to 10 kg. Plot the...
-
Water is being heated in a vertical piston-cylinder device. The piston has a mass of 40 kg and a cross-sectional area of 150 cm2. If the local atmospheric pressure is 100 kPa, determine the...
-
Water is boiled in a pan covered with a poorly fitting lid at a specified location. Heat is supplied to the pan by a 2 - kW resistance heater. The amount of water in the pan is observed to decrease...
-
1,600 Balance Sheet The following is a list (in random order) of KIP International Products Company's December 31, 2019, balance sheet accounts: Additional Paid-In Capital on Preferred Stock $2,000...
-
Question 3 4 pts 9 x + 3 x 9 if x 0 Find a) lim f(x), b) lim, f(x), C), lim , f(x) if they exist. 3 Edit View Insert Format Tools Table : 12pt M Paragraph B IV A2 Tv
-
Mr. Geoffrey Guo had a variety of transactions during the 2019 year. Determine the total taxable capital gains included in Mr. Guo's division B income. The transactions included: 1. On January 1,...

Study smarter with the SolutionInn App


