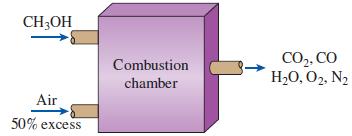
Methyl alcohol (CH 3 OH) is burned with 50 percent excess air. The combustion is incomplete with
Question:
Methyl alcohol (CH3OH) is burned with 50 percent excess air. The combustion is incomplete with 10 percent of the carbon in the fuel forming carbon monoxide. Calculate the mole fraction of carbon monoxide and the apparent molecular weight of the products.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:





