Ethane (C 2 H 6 ) is burned with 20 percent excess air during a combustion process.
Question:
Ethane (C2H6) is burned with 20 percent excess air during a combustion process. Assuming complete combustion and a total pressure of 100 kPa, determine
(a) The air–fuel ratio
(b) The dew-point temperature of the products.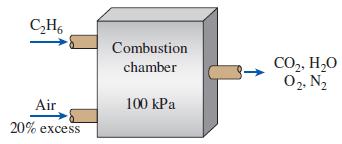
Transcribed Image Text:
C,H, Combustion CO,, H20 02, N2 chamber Air 100 kPa 20% excess
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
The fuel is burned completely with excess air The AF and the dew point of the products are to be det...View the full answer

Answered By

Mustafa olang
Please accept my enthusiastic application to solutionInn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group. For example, I created songs to teach my three-year-old campers the camp rules, but I gave my college student daily quizzes to help her prepare for exams.
I am passionate about helping students improve in all academic subjects. I still remember my excitement when my calculus student received her first “A” on a quiz! I am confident that my passion and experience are the qualities you are looking for at solutionInn. Thank you so much for your time and consideration.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Ethane (C2H6) is burned with 30% excess air during a combustion process. Assuming complete combustion and a total pressure of 100 kPa, determine. (a) The air-fuel ratio. (b) The dew point temperature...
-
Propylene (C3H6) is burned with 50 percent excess air during a combustion process. Assuming complete combustion and a total pressure of 105 kPa, determine (a) The airfuel ratio and (b) The...
-
Hydrogen (H2) at 7°C is burned with 20 percent excess air that is also at 7°C during an adiabatic steady-flow combustion process. Assuming complete combustion, determine the exit temperature...
-
The following is the stockholders' equity section from Chipotle Mexican Grill, Inc.'s balance sheet (in thousands, except per share data). a. Show the computation to derive the $375 thousand for...
-
A Calgary observatory records daily the mean counting rates for cosmic rays for that day. If a daily mean rate below 3200 is considered low, then "low" values occur about 3.6% of the time (in no...
-
. 4.(TCOs2,3,4)Oneofyourcorporateclientshasapproachedyouaboutwhetherornotitsemployeesarerequiredto...
-
Compare the mean silhouette values for the two cluster models. Which model is preferred?
-
A liquid mixture of 27 wt% acetone and 73 wt% water is to be separated at 25 o C into a raffinate and extract by multistage, steady-state, countercurrent liquid-liquid extraction with a solvent of...
-
3. (16) The French energy company Total SA announced yesterday that they were purchasing land and equipment for $1 billion to build additional wind farms and that they expected a 10% retum on that...
-
The following represents a project that should be scheduled using CPM: a. Draw the network. b. What is the critical path? c. What is the expected project completion time? d. What is the probability...
-
Methyl alcohol (CH 3 OH) is burned with the stoichiometric amount of air. Calculate the mole fractions of each of the products, and the apparent molar mass of the product gas. Also, calculate the...
-
Methyl alcohol (CH 3 OH) is burned with 50 percent excess air. The combustion is incomplete with 10 percent of the carbon in the fuel forming carbon monoxide. Calculate the mole fraction of carbon...
-
Return to the data in the preceding question. A year ago receivables stood at \($66,900\), and sales for the current year totaled \($706,600\). How many days did it take you to collect your average...
-
Use the following information for questions 1 and 2. Caterpillar Financial Services Corp. (a subsidiary of Caterpillar) and Sterling Construction sign a lease agreement dated January 1, 2020, that...
-
Identifying Binomial Distributions. Determine whether the given procedure results in a binomial distribution or a distribution that can be treated as binomial (by applying the 5% guideline for...
-
Case 6: TOMS Shoes in 2016: An Ongoing Dedication to Social Responsibility, by Margaret A. Peteraf, Sean Zhand, and Meghan L. Cooney (page C-57) Read the case and then respond to the case questions...
-
Quatro Co. issues bonds dated January 1, 2019, with a par value of $740,000. The bonds' annual contract rate is 13%, and interest is paid semiannually on June 30 and December 31. The bonds mature in...
-
Wildcat Mining wants to know the appropriate discount rate to use in their capital budgeting decision making process. Based on the following data, what is the weighted average cost of capital the CFO...
-
Answer each of the following question. What amount is 16 2/3% less than $66?
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
Air enters a 30-cm-diameter cooling section at 1 atm, 35oC, and 45 percent relative humidity at 18 m/s. Heat is removed from the air at a rate of 750kJ/min. Determine (a) The exit temperature (b) The...
-
Repeat Prob. 14-67 for a heat removal rate of 950kJ/min. Prob. 14-67 Air enters a 30-cm-diameter cooling section at 1 atm, 35oC, and 45 percent relative humidity at 18 m/s. Heat is removed from the...
-
A heating section consists of a 15-in-diameter duct that houses a 4-kW electric resistance heater. Air enters the heating section at 14.7 psia, 50oF, and 40 percent relative humidity at a velocity of...
-
A company manufactures lawnmowers. Compute the total amount of period costs from thr following costs.
-
TestAnswerSavedHelp opens in a new windowSave & ExitSubmit Item 1 7 1 0 points Time Remaining 1 hour 2 0 minutes 1 8 seconds 0 1 : 2 0 : 1 8 Item 1 7 Time Remaining 1 hour 2 0 minutes 1 8 seconds 0 1...
-
Use the following information for the Problems below. (Algo) [The following information applies to the questions displayed below.] Lansing Company's current-year income statement and selected balance...

Study smarter with the SolutionInn App


