Using Daltons law, show that for a real-gas mixture of k gases, where Z is the compressibility
Question:
Using Dalton’s law, show that
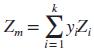
for a real-gas mixture of k gases, where Z is the compressibility factor.
Transcribed Image Text:
Z„ = EyZi %3D i=1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
Using Daltons law it is to be shown that for a realgas mixture Analysis U...View the full answer

Answered By

BETHUEL RUTTO
Hi! I am a Journalism and Mass Communication graduate; I have written many academic essays, including argumentative essays, research papers, and literary analysis. I have also proofread and written reviews, summaries and analyses on already finished works. I am eager to continue writing!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
A gas turbine is operated with gases (Cp = 0.992 kJ/kg K and = 1.29) entering it at 10 bar and 1025C, and leaving it at 1 bar and 550C. Assuming adiabatic flow through the turbine, calculate the...
-
A gas mixture at 300 K and 200 kPa consists of 1 kg of CO2 and 3 kg of CH4. Determine the partial pressure of each gas and the apparent molar mass of the gas mixture.
-
A mixture of Ne and Ar gases at 350 K contains twice as many moles of Ne as of Ar and has a total mass of 50.0 g. If the density of the mixture is 4.00 g/L, what is the partial pressure of Ne?
-
Lucy, a 57-year-old widow, lost her husband in 2021. He was an active participant in the Federal Employees' Retirement System (FERS), and his FERS contributions totaled $27,000 at the time of his...
-
In the previous question, 1350 is an outlier. If we remove this value, show why the sales without outliers are normally distributed at a 0.05 level of significance.
-
Using Wikpedia (www.wikpedia.com), look up the entry for Sarbanes-Oxley Act. Look over the table of contents and find the section that describes Section 404. What does Section 404 require of...
-
The face value of commercial paper borrowings at December 31,2020 , was \(\$ 6\) million. The six-month loan originated on September \(1,2020\). a. Prepare the journal entry for issuance of the loan...
-
Green Pastures is a 400-acre farm on the outskirts of the Kentucky Bluegrass, specializing in the boarding of broodmares and their foals. A recent economic downturn in the thoroughbred industry has...
-
NEED ANSWER WITH EXPLANATION. Q15 X Y A B C D E F G H K L M N Instructions To be completed i) Calculate the (Average Days Sales Outstanding Ratio) for 20XX(CY) & 20XX-1(PY) years and analyze any t...
-
Larry Power started a new business in the name of Power Electrical on October 1, 2020. During October, a number of activities occurred and the following totals resulted at October 31, 2020 (shown in...
-
A mixture of gases consists of 1 kmol of carbon dioxide, 1 kmol of nitrogen, and 0.3 kmol of oxygen. Determine the total amount of work required to compress this mixture isothermally from 10 kPa and...
-
A 60-L rigid tank contains an ideal-gas mixture of 5 g of N 2 and 5 g of CO 2 at a specified pressure and temperature. If N 2 were separated from the mixture and stored at mixture temperature and...
-
Eastern Polymers, Inc., processes a base chemical into plastic. Standard costs and actual costs for direct materials, direct labor, and factory overhead incurred for the manufacture of 23,500 units...
-
What are some inquiries an auditor can make when examining revenue and expense to add value to the audit?
-
What is meant by the phrase "dual dating of a report," and when would an auditor use this practice?
-
What is the auditor's responsibility for other information in the text portion of annual reports and other documents containing audited financial statements?
-
Why is an auditor concerned with a client's revenue recognition policies?
-
What is the auditor's responsibility for information that is supplementary to the financial statements but required by the FASB or GASB? How does the auditor report on such information?
-
Simulate the experiment described in Exercise 3.15, using any five identically shaped objects, two of which are one color and three another. Mix the objects, draw two, record the results, and then...
-
What did Lennox gain by integrating their WMS, TMS, and labor management systems?
-
A reversible absorption refrigerator consists of a reversible heat engine and a reversible refrigerator. The system removes heat from a cooled space at - 15oC at a rate of 70 kW. The refrigerator...
-
An ammonia-water absorption refrigeration cycle is used to keep a space at 25oF when the ambient temperature is 70oF. Pure ammonia enters the condenser at 300 psia and 140oF at a rate of 0.04lbm/s....
-
Describe the Seebeck and the Peltier effects.
-
Soru 4 L-1 e-5s (s - 36) *s) A hibiri =? u(t-5)*[((e^(30-6t))/72) + ((e^(6t-30))/72) - 1/36] Cu(t-5)*[((e^(25-t))/34) + (2*(e^(t-5))/27) - 7/3] BCD Du(t-5)*[((e^(3-4t))/6) + ((e^(2t-3))/12) - 1/6] E...
-
What is the major intracellular cation? a.Potassium b.Calcium c.Magnesium d.Sodium 2.What is the major extracellular cation? a.Sodium b.Chloride c.Magnesium d.Calcium 3.Osmolality can be defined as a...
-
Find the derivative. r = 14-08 cos 0 (dr/de) = 807 sin e (dr/de) = : 807 sin 0 - 08 cos e (dr/de) = : 807 cos 0 - 08 sin e (dr/de) == - 807 cos 0 + 08 sin e Question 22 Find dy/dx by implicit...

Study smarter with the SolutionInn App


