Many biologically important molecules are optically active. When linearly polarized light traverses a solution of compounds containing
Question:
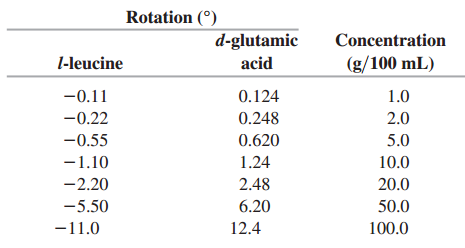
From these data, find the relationship between the concentration C (in grams per 100 mL) and the rotation of the polarization (in degrees) of each amino acid.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

University Physics with Modern Physics
ISBN: 978-0321696861
13th edition
Authors: Hugh D. Young, Roger A. Freedman, A. Lewis Ford
Question Posted:





