The following synthetic routes are incorrect. What is wrong witheach? (a) 1. Br2, CH3CO2H 2. Pyridine, heat
Question:
The following synthetic routes are incorrect. What is wrong witheach?
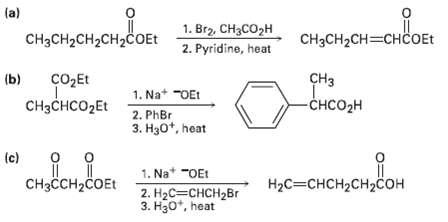
Transcribed Image Text:
(a) 1. Br2, CH3CO2H 2. Pyridine, heat CH3CH2CH2CH2COET CH3CH2CH=CHCOET (b) CHз CO2Et 1. Na* -OEt -CHCO2H CHзснсO2Et 2. PhBr 3. H30*, heat (c) 1. Na* "OEt Нас3снCH2сHсон CHзсCH2CОEt 2. НаС— снсH2Br 3. H30*, heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (14 reviews)
a Reaction with Br at the a position occurs only with al...View the full answer

Answered By

Umber Talat
I am providing full time mentoring and tutoring services in Business Finance, Contemporary issue in Global Economy, Quantitative Techniques, Principles of Marketing, strategic marketing, International Marketing, Organizational Behavior (OB), Consumer Behavior, Sales Force Management, Strategic Brand Management, Services Marketing, Integrated Marketing Communication (IMC), Principles of Management, General Management, Strategic Management, Small and Medium Enterprise Management, Innovation Management, Change Management, Knowledge Management, Strategic Planning, Operations Management, Supply Chain Management, Logistics Management, Inventory management, Total Quality Management (TQM), Productions Management, Project Management, Production Planning, Human Resource Management (HRM), Human Resource Development, Strategic HRM, Organizational Planning, Performance and Compensation Management, Recruitment and Selection, Organizational Development, Global Issues in Human Resource Management, Retail Marketing, Entrepreneurship, Entrepreneurial Marketing, International Business, Research Methods in Business, Business Communication, Business Ethics.
4.70+
158+ Reviews
236+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What is a synthetic lease? How are such leases structured, and what is their primary purpose? Is it likely that the use of synthetic leases will increase or decrease?
-
What is wrong with this "proof" that all horses are the same color? Let P(n) be the proposition that all the horses in a set of n horses are the same color. Inductive Step: Assume that P(k) is true,...
-
What is synthetic rating? What is its principal weakness?
-
Reference frame S is moving along the x axis at 0.6c relative to frame S. A particle that is originally at x = 10 m at t 1 = 0 is suddenly accelerated and then moves at a constant speed of c/3 in...
-
A friend in a distant city is considering moving to your area for more education and training in your field. Your friend has asked you for information about your program of study. Write a letter...
-
Find the output resistance for the active-cascode circuit of Fig. 3.77 excluding resistor R. Assume that all the transistors operate in the active region with dc drain currents of 100 A. Use the...
-
2. Prove that each of the following functions is uniformly continuous on (0,1). (a) (b) (c) (d) (e) x3 -1 f(x) = --1' x- . 1 f(x) = xsm-. x f(x) is any polynomial. f(x) = sin x . x
-
Neville Companys standard materials cost per unit of output is $10 (2 pounds _ $5). During July, the company purchases and uses 3,200 pounds of materials costing $16,160 in making 1,500 units of...
-
The poor w without.com BEMINDER. PO Oh your man to Use Question 1 Trucos condom Question 2 5 D
-
A firm needs to fully satisfy the demand, which is fixed to 900 units and generates unit revenue of $10. However, its production is subject to the random yield. That is, only a random percentage of...
-
The two isomers cis- and trans-4-tert-butyl-2-methylcyclohexanone are inter-converted by base treatment. Which isomer do you think is more stable, and why?
-
At tempted Grignard reaction of Cyclohexanone with tert-butyl magnesium bromide gives only about 1% yield of the expected addition product along with 99% un-reacted Cyclohexanone. If D 3 O + is added...
-
Discuss the similarities and differences between trend following and crosssectional momentum strategies.
-
Prove (11.32) . E (Yi,k | Zi = 0, = e) = E (Yi,k | i = 1, = e) = E (Yi,k | Ti = e), k = 1,2. (11.32)
-
University Medical Center needs to move from its existing facility to a new and larger facility five miles away from its current location. Due to construction delays, however, much of the new...
-
Calculate the base value or lump sum for each of the single and married filing jointly 2016 brackets given in Table 6.4. Table 6.4 ITABLE 6.4 Corporate Income Brackets and Tax Rates, 2015 Taxable...
-
Show that staged column diameter is proportional to (feed rate) \({ }^{1 / 2}\) and to \((1+\mathrm{L} / \mathrm{D})^{1 / 2}\).
-
An atmospheric column with 25 real stages is operating with a pressure drop of 0.6 in. of water per stage. Assume pressure drop in the condenser and the reboiler is \(1.2 \mathrm{in}\). of water...
-
Fire both employees and immediately implement a strict Internet and e-mail monitoring policy. You cannot risk possible charges of creating a hostile work environment because people are viewing...
-
Calculate the Lagrange polynomial P 2 (x) for the values (1.00) = 1.0000, (1.02) = 0.9888, (1.04) = 0.9784 of the gamma function [(24) in App. A3.1] and from it approximations of (1.01) and (1.03).
-
Predict the geometry and bond angles of PCl 3 .
-
(a) Using the principle of microscopic reversibility, give a detailed mechanism for the acid-catalyzed hydrolysis of methyl benzoate (structure in Eq. 20.16, p. 965) to benzoic acid and methanol. (b)...
-
Give the structure of the ester formed when (a) Succinic acid reacts with a large excess of diazomethane in ether. (b) Benzoic acid reacts with allyl bromide and K2CO3 in acetone.
-
Which one of the following compounds forms a cyclic anhydride on heating: methylmalonic acid or 2,3-dimethylbutanedioic acid?
-
A government bond matures in 30 years, makes semi-annual coupon payments of 6.0% ($120 per year) and offers a yield of 3.7% annually compounded. Assume face value is $1,000. Three years later the...
-
Your objective is: 1. Carry out a life insurance needs analysis, for each one of them (show your calculations) [30 Marks] 2. Refer to the case and the insurance plan quotes. Would you recommend...
-
TufStuff, Incorporated, sells a wide range of drums, bins, boxes, and other containers that are used in the chemical industry. One of the company s products is a heavy - duty corrosion - resistant...

Study smarter with the SolutionInn App


