Although aldehydes and ketones are weak acids, their a-hydrogens are rnore than 30 pKa units more acidic
Question:
Although aldehydes and ketones are weak acids, their a-hydrogens are rnore than 30 pKa units more acidic than the hydrogen's of alkanes.
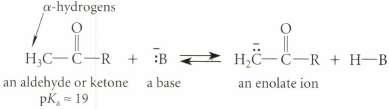
Using polar effects and resonance effects in your argument, explain the enhanced acidity of aldehydes and ketones.
Transcribed Image Text:
α-hydrogens an aldehyde or ketone pK 19 a base an enolate ion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
The conjugatebase enolate ion is stabilized by the polar electro...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Name the following aldehydes and ketones according to IUPAC rules: (c) (a) , (b) CCH2 CHCH2CHCH C CHCCH2CH2H2H-CH (e) CH (f) (d) "op CH;CH3CHCH,C,H , * " "CH
-
Acetal formation is a characteristic reaction of aldehydes and ketones, but not of carboxylic acids. Show how you could advantageously use a cyclic acetal protecting group in the following synthesis:...
-
Explain why aldehydes and ketones react with a weak acid such as hydrogen cyanide in the presence of -CN, but do not react with strong acids such as HCl or H2SO4 in the presence Cl- or HSO4-.
-
The following table includes the GNP(X) and the demand for food (Y) for a country over ten years period. Year 1980 1981 1982 1983 1984 1985 1986 1987 1988 1989 Y 6 7 8 10 8 9 10 9 11 10 X 455555 50...
-
Jims Landscaping is in the business of maintaining and improving yards in surrounding areas. The company bases its overhead cost budgets on the following data: Variable:Overhead costs Supplies...
-
The total assets and liabilities of Omni Company at January 1 and December 31, 2012, are presented below. Determine the amount of net income or loss for 2012, applying each of the following...
-
a. Repeat Problem 15.H1 (use the Maxwell-Stefan equations), but bulb 1 at \(\mathrm{z}=0\) contains the following mole fractions: \(\mathrm{y}_{\text {air }}=0.500, \mathrm{y}_{\mathrm{H} 2}=0.500\),...
-
Bunker Company negotiated a lease with Gilbreth Company that begins on January 1, 2014. The lease term is three years, and the assets economic life is four years. The annual lease payments are...
-
Based on the preceding results, is the sample median an unbiased estimator of the population median? Why or why not? A. The sample median targets the population median. As such, the sample median...
-
The file Problem5_9.xlsx contains passing statistics for NFL teams during the 2017 season. Using formulas that do combine INDEX and MATCH in the same cell, return the desired statistics in the range...
-
Explain why borazole (sometimes called inorganic benzene) is a very stable compound. N:N borazole
-
Which of the following two alkyl halides would react most rapidly in a solvolysis reaction by the SN1 mechanism? Explain your reasoning. Ch 1,0-CH-CH-CH,-Cl A (trans isomer) CH.O-C-CH,-CI CH2
-
Peter Rourke, a loan processor at Royal Bank, has been timed performing four work elements, with the results shown in the following table. The allowances for tasks such as this are personal, 7%;...
-
Thunder Corporation, an amusement park, is considering a capital investment in a new exhibit. The exhibit would cost $144,123 and have an estimated useful life of 7 years. It can be sold for $68,000...
-
(a) Calculate the absolute pressure at the bottom of a fresh-water lake at a depth of 24.0 m. Assume the density of the water is 1.00 10 3 kg/m 3 and the air above is at a pressure of 101.3 kPa. Pa...
-
Determine the R value and U-factor for the following assemblies.Assume winter conditions. Use the attached Tables. Exterior Wall Component R-Value 3/8\" Plywood 1\" Air Space 3 1/2\" Fiberglass Batt...
-
DISCUSS THE CONCEPT OF Change Management.show the barriers of organizational change
-
The following information has been extracted from the records of Mas Bhd. about one of its products "LITTLE JOMBO". Mas Bhd. uses the perpetual inventory system and its reporting period ends on 30...
-
Evaluate the given expressions all numbers are approximate. (26.5) 2 (9.85) 3
-
Why can wastewater treatment requirements in Hawaii be less stringent than those in most locations on the U.S. mainland?
-
The following reaction, called the benzilic acid rearrangement, takes place by typical carbonyl-group reactions. Propose a mechanism (Ph =phenyl). 1. NaOH, H,0 2. H30+ Ph C-C Ph Ph Ph Benzil...
-
The step-growth polymer nylon 6 is prepared from caprolactam. The reaction involves initial reaction of caprolactam with water to give an intermediate open?chain amino acid, followed by healing to...
-
Qiana, a polyamide fiber with a silky texture, has the following structure. What is the monomer units used in the synthesis ofQiana? flomdoCor CH2- Qiana CICH2)6C-NH- NH-
-
Kate owns 25 real estate rental properties that she manages herself. She averages about 20 hours a week taking care of her properties. This year, the rental properties produced a loss of $35,000....
-
Consider the following linear programming problem: Max x1+2x2 s.t. x1+x2 <3 x12x20 x21 x1, x20 a. Identify the feasible region. (12 points) b. Are any of the constraints redundant? If yes, then...
-
On December 31, 2023, Tyler Partnership reported a $68,000 loss on its books. The items included in the loss computation were $21,000 in sales revenue, $15,000 in dividends, $21,000 in cost of goods...

Study smarter with the SolutionInn App


