Amino acids, from which proteins are formed, exist as ?dipolar ions.? The structure of the dipolar ion
Question:
Amino acids, from which proteins are formed, exist as ?dipolar ions.? The structure of the dipolar ion of the amino acid alanine is
(a) Calculate the formal charges on all of the atoms, except hydrogen?s of alanine.
(b) What is the overall charge of alanine?
(c) Explain whether or not you expect the two oxygen?s to be different.
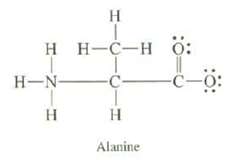
Transcribed Image Text:
H T HH-C-HÖ: H-N- H -C- I H Alanine -C-0:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 86% (15 reviews)
a b The overall charge of ala...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Calculate the formal charges on all of the atoms except hydrogen's, in these compounds: a) H-N-N=N: c) H-C-N=N: H e) H H-=C-H b) H-N-N-N: HO: H-C-C-C 1 H d) f) H H-B-H T H
-
Explain whether or not the following particles have an acceleration: (a) A particle moving in a straight line with constant speed and (b) A particle moving around a curve with constant speed.
-
In the previous exercise, explain whether or not you believe the sample is representative of the population of interest and why. In order to investigate how many hours a day students at their school...
-
The Illinois State University Alumni Association is having two Baseball Day events this year. The purpose of the events is to both foster relationships between ISU alums and to also raise money for...
-
The Real life highlights the risks to manufacturers of requiring suppliers to provide inventory on a just-in-time basis. In your own words, explain the advantages and disadvantages ix suppliers if...
-
What is an initial margin requirement? If its value is 0.50, and if you have $10,000 to invest you can buy securities with a total market value of $______.
-
Lake Company has the following shares outstanding: 20,000 shares of \(\$ 50\) par value, five percent cumulative preferred stock and 80,000 shares of \(\$ 10\) par value common stock. The company...
-
The controller of a retail company has just had a $50,000 request to implement and ABC system quickly turned down. A senior vice president, in rejecting the request, noted. Given a choice, I will...
-
Assume a firm has earnings before depreciation and taxes of $470,000 and depreciation of $170,000. if it is an 35 percent tax bracket , compute its cash flow. if its in a 20 percent tax bracket ,...
-
Globalization and technology developments have led to some significant changes in the global economy. Discuss the importance of such changes to the United States. What is the impact upon supply...
-
Show a Lewis structure for NO 2 , both oxygens are bonded to the nitrogen.) Show a resonance structure also.
-
A covalent ion can also have polar bonds. Consider the ammonium caution. I low arc its bonds polarized? Do you think that the N of the ammonium caution is more or less ?electro negative? than the N...
-
What are two important qualities of evidence that increase its reliability?
-
In the peaceful town of Oakwood, Sweet Delights, a beloved bakery renowned for its delectable treats, found itself entangled in an unintentional risk that would test the principles of tort law. The...
-
If y+xy-3x= 9, and dy dt dx = -1 when x = 1 and y = -4, what is when x = = 1 and y = -4? dt dx dt
-
Margaret Lewis was a tenured teacher in a suburban school district. After work one day, she went to happy hour with her colleagues and had several glasses of wine. On her way home, she was stopped by...
-
Monica Gellar owns a famous restaurant called Famous Kitchen. During a busy night at the restaurant, a spark went off in the kitchen and caused a fire. The restaurant had to immediately shut down....
-
Shaw Drumgold was convicted and imprisoned for murder of a 12-year old girl. The key witness against him, Ricky Evans, a homeless man, was given cash, meals and housing at a local hotel for several...
-
At the final exam in a statistics class, the professor asks each student to indicate how many hours he or she studied for the exam. After grading the exam, the professor computes the least-squares...
-
(a) With not more than 300 words, examine why Malaysia missed its 2020 targets to divert 40% of waste from landfills and increase recycling rates to 22%. (10 marks) b) Why arent Malaysian recycling?...
-
Graph the function f(x) = sin 50x in an appropriate viewing rectangle.
-
Of the two vitamins A and C, and is hydrophilic and water-soluble while the other is hydrophobic and fat-soluble. Which iswhich? CH CH- CH CH2OH H3C CH3 H- CH Vitamin A (retinol) Vitamin C...
-
Fill in the multiple bonds in the following model of naphthalene, C10H8 (gray = C, ivory =H). How many resonance structures does naphthalenehave?
-
The following model is a representation of ibuprofen, a common over-the-counter pain reliever. Indicate the positions of the multiple bonds, and draw a skeletal structure (gray = C, red = O, ivory...
-
Math 10 - February 8, 2018Quiz 3-1Name______Answer Key_____________Time: 30 MinutesNote: Do not share calculator or material among each other. Question 1 (5 points)The time (in minutes) until the...
-
Christy General Hospital, a nonprofit organization, had the following transactions during its fiscal year. 1. The hospital received a gift of $540,000 in equity securities from Brady Johnson. The...
-
Code Sec. 409(p) prescribes that a "non-allocation year" generally means an ESOP plan year during which on any date disqualified persons own _________________ of the stock in the S corporation. a. 25...

Study smarter with the SolutionInn App


