Assign R or S configuration to the following molecule, write the product you would expect from SN2
Question:
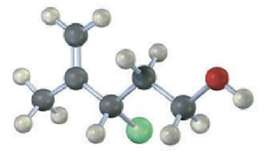
Assign R or S configuration to the following molecule, write the product you would expect from SN2 reaction with NaCN, and assign R or S configuration to the product (yellow-green =Cl):
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
CH HH CHOH NaCN H3C CH HH R NC H CH2OH NaCl HCI T...View the full answer

Answered By

Ashish Bhalla
I have 12 years work experience as Professor for Accounting, Finance and Business related subjects also working as Online Tutor from last 8 years with highly decentralized organizations. I had obtained a B.Com, M.Com, MBA (Finance & Marketing). My research interest areas are Banking Problem & Investment Management. I am highly articulate and effective communicator with excellent team-building and interpersonal skills; work well with individuals at all levels.
4.80+
17+ Reviews
46+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assign R or S configuration to the chirality center in the following molecular model of the amino acid methionine (blue = N, yellow =S):
-
Assign R or S configuration to the chirality center in each of the followingmolecules: CH (c) . (b) (a) 2 "- HS efpo -- CH-
-
Assign R or S configuration to each chirality center in the followingmolecules: (a) (b) ,CH (c) , . CH CHCH
-
In the Walton Bookstore example with a discrete demand distribution, explain why an order quantity other than one of the possible demands cannot maximize the expected profit.
-
Evaluate Brian Conrad's approach to appraisal interviews. Write a paragraph or two summarizing what Conrad is doing well and how he might further improve the effort.
-
How could you keep your employees from going to your competitors after you have trained them?
-
Repeat Example 14-2 except for a single-stage system and unknown underflow product concentration. Example 14-2 We wish to treat 1000.0 kg/h (wet basis) of insoluble meal (D) that contains 20.0 wt%...
-
The trial balance of De Bortoli Co. shown below does not balance. Each of the listed accounts has a normal balance per the general ledger. An examination of the ledger and journal reveals the...
-
Match the following examples to the element of culture that they represent. Example Element of Culture Flickr wants to make sure its employees balance work and fun. If things in the office get too...
-
On August 31, 2021, Claresview Company had a cash balance per its books of $26,030. The bank statement on that date showed a balance of $17,100. A comparison of the bank statement with the Cash...
-
From what alkyl bromide was the following alkyl acetate made by SN2 reaction? Write the reaction, showing allstereochemistry.
-
Draw the structure and assign Z or F stereochemistry to the product you expect from E2 reaction of the following molecule with NaOH (yellow-green =Cl):
-
You completed a managerial accounting class last semester and learned about budgeting concepts. How do government budgeting concepts differ from those used in a corporate setting?
-
Garcia Company sells snowboards. Each snowboard requires direct materials of $110, direct labor of $40, variable overhead of $55, and variable selling, general, and administrative costs of $13. The...
-
(a) profits will be maximized at a level of output of units, and (b) at this level of output, profits will be $_____. (Show your calculations.) (c) Is this firm operating in a perfectly competitive...
-
A firm is considering three projects: Project 1, project 2, and Project 3. Capital requirements and budget limitations in the next three years prevent the firm from undertaking all of these at this...
-
Western Athletic Club International ( WACI ) owns and operates a chain of fitness clubs and is interested in estimating the CLV for new memberships. Practically all of WACI's costs are fixed costs....
-
Lethe has borrowed $100 000 to purchase a drilling machine and promise to make 5 years of annual repayments at a fixed interest rate of 11.5% p.a. What is the annual repayment?
-
Firm A operates in a B2B industry. Firm B operates in a business to consumer (B2C) industry. Which firm is likely to have lower accounts receivable as a percentage of its sales?
-
Rosalie owns 50% of the outstanding stock of Salmon Corporation. In a qualifying stock redemption, Salmon distributes $80,000 to Rosalie in exchange for one-half of her shares, which have a basis of...
-
Consider the reaction: If a solution initially contains 0.210 M HC 2 H 3 O 2 , what is the equilibrium concentration of H 3 O + at 25 C? HCH3O (aq) + HO(1) H3O+(aq) + CH3O (aq) K 1.8 x 10-5 at 25 C =
-
Draw all four resonance forms of the fragment at m/z 73 in the mass spectrum of pentanoic acid.
-
Name the following carboxylic acid derivatives, giving both a common name and an IUPAC name where possible. (a) PhCOOCH2CH(CH3)2 (b) PhOCHO (c) PhCH(CH3) COOCH3 (d) PhNHCOCH2CH(CH3)2 (e) CH3CONHCH2Ph...
-
(a) Show how you would use acetic anhydride and an appropriate alcohol or amine to synthesize (i) benzyl acetate, (ii) N,N-diethylacetamide. (b) Propose a mechanism for each synthesis in part (a).
-
Sony of Japan produces DVD players and exports them to the United States . Last year the exchange rate was 130 / $ and Sony charged $ 150 per DVD player . Currently the spot exchange rate is 110 / $...
-
Hi, I have various models (train and test results) created out of Logistic regression, RF and LDA. How do I store the accuracy, precision, and recalls values into a dataframe ? Thanks, Bala
-
Estimate a logistic regression using "buyer" as the dependent variable and the following as predictor variables: last_purch dollars customer_type customer_sqft cab_franc cab_sauvignon malbec merlot...

Study smarter with the SolutionInn App


