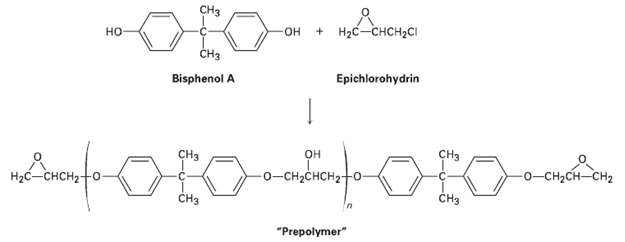
Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with
Question:
Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with epichiorohydrin forms a ?prepolymer,? which is then ?cured? by treatment with a triamine such as H2NCH2CH2NHCH,CH2NH2,. Draw structures to show how addition of the triamine results in strengthening he polymer. Amines are good nucleophiles and can open epoxide rings in the same way other bases can.
Transcribed Image Text:
CH3 Аоне OLO- + Hас-снсн-с но -он ČH3 Epichlorohydrin Bisphenol A CHз CHз н Hас — снсн?- -о-сн-снсH2 -о-сн-сH—сH2 CHз CHз "Prepolymer"
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Addition of the triamine causes ...View the full answer

Answered By

Talha Talib
I am a member of IEEE society. As i am a student of electrical engineering badge 17 but beside of this i am also a tutor in unique academy. I teach calculus, communication skills, mechanics and economics. I am also a home tutor. My student Muhammad Salman Alvi is a brilliant A-level student and he performs very well in academics when i start to teach him. His weak point was mathematics but now he is performing well in mathematics. I am a scholarship holder in Fsc as i scored 1017 marks in metric out of 1100. Later on i got scholarship in Punjab Group of Colleges. I got 2nd position in robotics competition in 2018 as my project home automation select for the exhibition in Expocentre.
4.60+
23+ Reviews
62+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Add curved arrows to the following structures to show how electron pairs must be moved to interconvert the structures, and locate any formal charges. O-H -H
-
Which SN2 reaction of each pair would you expect to take place more rapidly in a protic solvent? (a) (1) Or (2) (b) (1) Or (2) (c) (1) Or (2) (d) (1) Or (2) Br (1.0M)-MeO(1.0M)-. / OMe + Br
-
The S N 2 reaction between a Grignard reagent and an epoxide works reasonably well when the epoxide is ethylene oxide. However, when the epoxide is substituted with groups that provide steric...
-
Marty owns an auto body repair shop. The business has no real value without him and the building is pretty much a mess after being full of paint fumes for all those years but the land is worth...
-
Briefly compare the advantages and disadvantages of illustrating data with charts (bar and line) versus tables.
-
Explain why individual brands may not follow the product life-cycle pattern. Give an example of a new brand that is not entering the life cycle at the market introduction stage.
-
Consider the following financial statements for Benjamin Company. During 2019, management obtained additional bond financing to enlarge its production facilities. The company faced higher production...
-
Perkins Company acquired 100% of Schultz Company on January 1, 2010, for $161,500. On December 31, 2010, the companies prepared the following trial balances: Required: A. What method is being used by...
-
y = - 5 sin ( 8 x + pi / 2 ) + 7
-
Joe Schreiner, controller for On Time Clock Company Inc., recently prepared the company?s income statement and statement of changes in equity for 2020. Schreiner believes that the statements are a...
-
Propose structures for compounds with the following 1H NMR spectra: (a) C 5 H 9 ClO 2 ? ?IR: 1735 cm ?1 ? (b) C 7 H 12 O 4 ? ? ?IR: 1735 cm ?1 ? (c) C 11 H 12 O 2 ? ??IR: 1710 cm ?1 ? TMS 8. 6. O ppm...
-
In the iodoform reaction, a triiodomethyl ketone reacts with aqueous NaOH to yield a carboxylate ion and iodoform (triiodomethane). Propose a mechanism for thisreaction. OH H20 Cl3 HCI3 R.
-
What is the term for the right of Roche citizens to walk across Shoepkes land on the trail? Vern Shoepke purchased a two-story home from Walter and Eliza Bruster in the town of Roche, Maine. The...
-
Find the Sources and Uses of Funds Cash 70,000 67,200 Marketable Securities 304,000 310,000 Net Receivables 248,000 40,000 Inventories 8,400 9,200 Total Current Assets 630,400 426,400 Property and...
-
Here are data regarding Acme Company for the current year: Work in process inventory, beginning $10,500 Work in process inventory, ending $19,000 Cost of goods manufactured $333,500 Direct materials...
-
. As shown in fig.1, the frame supports the cylinder, its gravity is G. Determine the constraint force on the fixed hinged support A and B. AD=DC=3R, the radius of the pulley C is R. (20 marks) 450 D...
-
In the fundamental principles of a free market system, what is true of the equilibrium price? Explain in detail
-
Oil flows through the 100-mm-diameter pipe with a velocity of 5 m/s. If the pressure in the pipe at A and B is 80 kpa, determine the x and y components of force the flow exerts on the elbow. The flow...
-
What are the dimensions of the following quantities? (a) Coefficient multiplying \(x^{3}\) in nonlinear spring stiffness, \(k_{3}\) (b) The perturbation parameter, \(\varepsilon\) (c) A detuning...
-
A condenser (heat exchanger) brings 1 kg/s water flow at 10 kPa quality 95% to saturated liquid at 10 kPa, as shown in Fig. P4.91. The cooling is done by lake water at 20C that returns to the lake at...
-
What is a chemical bond according to MO theory?
-
Using benzene and any other reagents, outline a synthesis of each of the following compounds. Tert-butylcyclohexane
-
Give the products expected (if any) when nitrobenzene reacts under the following conditions. (a) Cl2, FeCl3, heat (b) Fuming HNO3, H2SO4 (c) H3C-C-CI, AICI,( 1.1 equiv.), then H2O
-
Explain how you would distinguish between ethylbenzene, p-xylene, and styrene solely by NMR spectroscopy.
-
Why are histograms important for research? They are useful for showing the data's relationship to the mean. They are most useful for qualitative data. They are only used with data that are skewed....
-
A company is considering the purchase of a new machine. The machine will cost $60,000 plus $25,000 for shipping and installation and falls under the 3-year MACRS class. Net Working Capital will rise...
-
A $ 5 comma 0 0 0 bond with a coupon rate of 6 . 2 % paid semiannually has two years to maturity and a yield to maturity of 6 . 9 % . If interest rates fall and the yield to maturity decreases by 0 ....

Study smarter with the SolutionInn App


