Explain whether the methyl is axial or equatorial in this compound: CH3 H C-C-CH3 CH3
Question:
Explain whether the methyl is axial or equatorial in this compound:
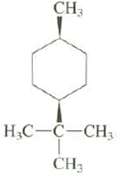
Transcribed Image Text:
CH3 H C-C-CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (13 reviews)
The axial destabilization energy of the ...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
The compound below is an example of a methyl ester. Methyl esters react with lithium iodide to give lithium carboxylate salts. The solvent in this example is pyridine (margin). Suggest several...
-
Explain why each compound or ion should be aromatic, antiaromatic, or nonaromatic. (a) The cyclonona-tetraene cation (b) The cyclononatetraene anion (c) The [16]annulene dianion (d) The [18]annulene...
-
The firm is considering between two mutually exclusive projects, Project A and Project B, that each require an upfront investment of $100m and are expected to produce free cash flow (FCF) only at the...
-
Predetermined plantwide overhead rate; alternative cost drivers Repeat the requirements for Exercise 7.22, assuming that Facsimile uses the following as overhead cost drivers: (a) Direct labour...
-
A national catalogue and Internet retailer has three warehouses and three major distribution centers located around the country. Normally, items are shipped directly from the warehouses to the...
-
On the basis of what you have learned about adjustments, why do you think that adjusting entries are made on the last day of the accounting period rather than at several times during the accounting...
-
Zimmerman Company of Shawnee, Kansas, spreads herbicides and applies liquid fertilizer for local farmers. On May 31, 2014, the companys Cash account per its general ledger showed a balance of...
-
Only need help with D,E,F Thank you! D:What would the stock price be if its dividends were expected to have zero growth? (D 0 = $2.00) E:Now assume that Temp Que is expected to experience supernormal...
-
The first column shows the number of donuts consumed, while the second and third columns (respectively) show Shanes and Miriams marginal personal use values. Assume that Shanes initial endowment is...
-
Draw both chair conformations of 1-methyl-1-phenylcyclohexane. Which is more stable by how much energy?
-
Draw both chair conformations for menthol (a component of peppermint oil) and its stereo isomer, neo menthol. Which groups are axial and which groups are equatorial? Explain which conformation is...
-
True or False IPO charts provide only brief descriptions of a functions input, processing, and output, but do not show the specific steps taken in a function.
-
Direct labor-hours Estimated Data Manufacturing overhead Direct labor-hours Machine-hours Job A Machine-hours Machining $ 4,212,000 Assembly $ 324,000 18,000 234,000 234,000 13,000 Machining Assembly...
-
Why don't governments like private issues of money, or even foreign currency, being used in their countries, and try to monopolise the issue of domestic money?
-
Prepare the journal entries for the following transactions: June 1 4 - Sold 6 units of inventory to Phil Connors. The sales price was $ 6 0 per unit and the cost of the units was $ 4 0 per unit. The...
-
Innovative Technologies Inc. assembles circuit boards by using a manually operated machine to insert electronic components. The original cost of the machine is $ 7 5 , 0 0 0 , the accumulated...
-
How is finance disrupted with this ecosystem? Explain briefly
-
\( \qquad \) refers to a company pulling revenues into the distribution channel fraudulently. a. Constructive intent b. Channel stuffing c. Gaming strategy d. Big bath e. Double play
-
Could a set of three vectors in span all of? Explain. What about n vectors in when n is less than m? R4
-
Find (/g)'(1) if f (1) = '(1) = g(1) = 2 and g'(1) = 4.
-
Qiana, a polyamide fiber with a silky texture, has the following structure. What is the monomer units used in the synthesis ofQiana? flomdoCor CH2- Qiana CICH2)6C-NH- NH-
-
What is the structure of the polymer produced by treatment of ?-propiolactone with a small amount of hydroxide ion? B-Propiolactone
-
Polyimides having the structure shown arc used as coatings on glass and plastics to improve scratch resistance how would you synthesize a polyimide? A polyimide -N-
-
Quinn was hired as the Chief Operating Officer (COO) of Zeta Corporation, a publicly-traded company in the technology sector, on April 1, 2022. For the fiscal year 2023, Zeta Corporation compensated...
-
Mind Challenge, Incorporated, publishes innovative science textbooks for public schools. The company's management recently acquired the following two new pieces of equipment. Computer - controlled...
-
FederalWay, Incorporated, is a major department store chain. The dominant portion of the company's business consists of providing merchandise and services to consumers through department stores and...

Study smarter with the SolutionInn App


