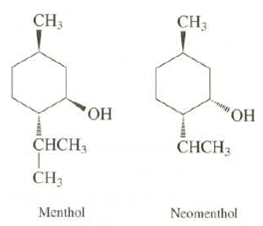
Draw both chair conformations for menthol (a component of peppermint oil) and its stereo isomer, neo menthol.
Question:
Draw both chair conformations for menthol (a component of peppermint oil) and its stereo isomer, neo menthol. Which groups are axial and which groups are equatorial? Explain which conformation is more stable for each stereo isomer. Which stereo isomer is more stable by how much energy?
Transcribed Image Text:
CH3 CH3 "ОН "ОН СНCH СНCH, CH3 Menthol Neomenthol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
For menthol the groups are either all axial or all equatorial Obviously th...View the full answer

Answered By

Ankit Mahajan
I am an electrical engineering graduate from Thapar institute of engineering and technology.
Qualified exams - GATE 2019,2020.
CAT EXAM 2021- 91.4 percentile
SSC EXAMS- 2019,2020,2021
AFCAT EXAM- 2019,2020,2021
I want to share my knowledge with other people so that they can achieve the same.
I have strong hold Mathematics, Electrical engineering and all the subjects related.
Just give me a problem and I will give you the solution of it.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw both chair conformations for each of the following compounds: (a) (b) (c) (d) (e) NH2
-
Draw both chair conformations for each of the following compounds. In each case, identify the more stable chair conformation: (a) Methylcyclohexane (b) Trans-1,2-Diisopropylcyclohexane (c)...
-
Draw both chair conformations of 1-methyl-1-phenylcyclohexane. Which is more stable by how much energy?
-
M DO NOT TURN THIS PAGE UNTIL YOU ARE TOLD TO DO SO SECTION B. ANSWER ANY THREE (3) QUESTIONS Question 1 (25 marks) You have analyzed Solar Power Ltd. and have reported the results of your analysis...
-
The following annual data relate to Facsimile Printing Pty Ltd: Budgeted machine hours............................15,000 Budgeted direct labour hours.......................30,000 Budgeted direct...
-
Anna Bellatorre, Inc. is interested in using its activity-based costing system to improve its operating efficiency and its profit margins by applying activity-based management techniques. As part of...
-
How might a company's mission statement affect how it does its business? Find Bass Pro's mission statement on its Web site [www.basspro.com]. (Hint: Look at the Newsroom link.) How does Bass Pro...
-
Freelin Conn filed a voluntary petition under Chapter 7 of the Bankruptcy Code on September 30, 2011. Conn listed Banc- Ohio National Bank as having a claim incurred in October 2010 in the amount of...
-
Fuivalent Units of Production and Related Costs The charges to Work in Process Assembly Department for a period, together with information concerning production, are as follows. All direct materials...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
Explain whether the methyl is axial or equatorial in this compound: CH3 H C-C-CH3 CH3
-
Draw the structures of these compounds. (a) (E)-3-Hexene (b) (Z)-2-Chloro-3-isopropyl-2-heptene
-
Harper, Inc., acquires 40 percent of the outstanding voting stock of Kinman Company on January 1, 2020, for $210,000 in cash. The book value of Kinmans net assets on that date was $400,000, although...
-
Activity 1.4: When Less Becomes More For this activity, refer to the images shown. This is an activity which was performed for you if you do not have available two identical mirrors at home. But if...
-
! Required information [The following information applies to the questions displayed below.] Aces Incorporated, a manufacturer of tennis rackets, began operations this year. The company produced...
-
During the early part of winter, one morning, two hunters decided to go quail hunting on a property where the owner had given them permission to hunt. A nearby forest ranger saw the hunters and...
-
Required information [The following information applies to the questions displayed below.] Trini Company set the following standard costs per unit for its single product. Direct materials (30 pounds...
-
A horticulturist knows that the weights of honeybees that have previously visited her orchard are normally distributed with a mean of 0.87 grams, and a population standard deviation of 0.15 grams....
-
In the example of planning a new menu, what do you think of the manager s plan? What problems might there be? If you were the manager, would you have people participate even more, such as visiting...
-
Define the essential properties of the following types of operating systems: a. Batch b. Interactive c. Time sharing d. Real time e. Network f. Parallel g. Distributed h. Clustered i. Handheld
-
What is the slope of the tangent line through the point (2, (2)) if '(x) = x 3 ?
-
How would you distinguish spectroscopically between the following isomer pairs? Tell what differences you would expect to see. (a) N-Methylpropanamide and N, N-dimethylacet amide (b)...
-
Propose a structure for a compound, C4H7ClO2 that has the following 1R and 1H NMRspectra: 100 80 60 40 20 4000 3000 2500 2000 1000 500 3500 1500 Wavenumber (cm-1) TMS O ppm 10 6. Chemical shift (8)...
-
Assign structures to compounds with the following 1H NMR spectra:? (a) C 4 H 7 ClO? ??IR: 1810 cm ?1 ? (b) C 5 H 7 NO 2 ? ?IR: 2250, 1735 cm ?1 ? (c) C 5 H 10 O 2 ? ? ?IR: 1735 cm ?1 ? TMS O ppm 10...
-
. Emerson Cammack wishes to purchase an annuity contract that will pay him $7,000 a year for the rest of his life. The Philo Life Insurance Company figures that his life expectancy is 20 years, based...
-
Integrity Inc. can sell 20-year, $1,000 par value bonds paying semi-annual interests with a 10% coupon. The bonds can be sold for $1,050 each; flotation cost of $50 per bond will be incurred in this...
-
Duncan Inc. issued 500, $1,200, 8%, 25 year bonds on January 1, 2020, at 102. Interest is payable on January 1. Duncan uses straight-line amortization for bond discounts or premiums. INSTRUCTIONS:...

Study smarter with the SolutionInn App


