Question: Natural gas containing a mixture of methane ethane propane and butane is burned in a furnace with excess air. (a) One hundred k mol/h of
Natural gas containing a mixture of methane ethane propane and butane is burned in a furnace with excess air.
(a) One hundred k mol/h of a gas containing 94.4 mole% methane, 3.40% ethane 0.60% propane, and 0.50% butane is to be burned with 17% excess air. Calculate the required molar flow rates of the air.
(b) Let nf(k mol/h) = molar flow of the fuel gas x1, x2, x3, x4, = mole fractions of methane, ethane, propane, and butane, respectively, in the fuel Pxs = percent excess air na (k mol/h) = molar flow rate of the air fed to the furnace Derive an expression for ha in terms of the other variables. Check your formula with the results of part (a).
(c) Suppose the feed rate and composition of the fuel gas are subject to periodic variations, and a process control computer is to be used to adjust the flow rate of air to maintain a constant percentage excess. A calibrated electronic flow meter in the fuel gas line transmits a signal R that is directly proportional to the flow rate (nf = aRf), with a flow rate of 75.0 k mol/h yielding a signal Rf = 60. The fuel gas composition is obtained with an on-line gas chromatograph. A sample of the gas is injected into the gas chromatograph (GC), and signals A1, A2, A3, and A4, which are directly proportional to the moles of methane, ethane, propane, and butane, respectively, in the sample, are transmitted. (Assume the same proportionality constant for all species.) The control computer processes these data to determine the required air flow rate and then sends a signal R1 to a control valve in the air line. The relationship between Ra and the resulting air flow rate, h, is another direct proportionality, with a signal R1 = 25 leading to an air flow rate of 550 k mol/h. Write a spreadsheet or computer program to perform the following tasks:
(i) Take as input the desired percentage excess and values of Rf, A1, A2, A3, and A4.
(ii) Calculate and print out nf, x1, x2, x3, x4, na, and Ra.
Test your program on the data given below assuming that 15% excess air is required in all cases. Then explore the effects of variations in Pxs and R1 on na for the values of A1 ? A4 given the third line of the data table. Briefly explain your results.?
(d) Finally, suppose that when the system is operating as described, stack gas analysis indicates that the air feed rate is consistently too high to achieve the specified percentage excess. Give several possible explanations.
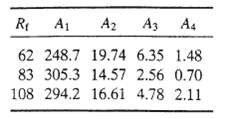
R A A2 A3 A4 62 248.7 19.74 6.35 1.48 83 305.3 14.57 2.56 0.70 108 294.2 16.61 4.78 2.11
Step by Step Solution
3.46 Rating (159 Votes )
There are 3 Steps involved in it
a CH4 20 CO 2HO CH60 2CO 3HO C3Hg 50 3CO 4HO 13 CH0 120 4CO 5H0 0944100kmol CH4 2 kmol O h 1 kmol CH ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
13-E-C-E-C-P (165).pdf
180 KBs PDF File
13-E-C-E-C-P (165).docx
120 KBs Word File


