Ribose, an essential part of ribonucleic acid (RNA), has the following structure: (a) How many chirality centers
Question:
Ribose, an essential part of ribonucleic acid (RNA), has the following structure:
(a) How many chirality centers does ribose have? Identify them.
(b) How many stereo isomers of ribose are there?
(c) Draw the structure of the enantiomer of ribose.
(d) Draw the structure of a diastereomer ofribose.
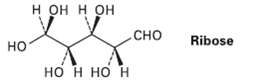
Transcribed Image Text:
н он н он Ribose но сно но н но н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
a c HO Ribose HO Ribose has three chirality centers whic...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many chirality centers are contained in (a) The aldotetrose (b) The ketopentose just given? (c) How many stereoisomers would you expect from each general structure?
-
How many chirality centers are present in estradiol how many stereo isomers does estradiol have?
-
Budgeting is not only an essential part of planning; but it also helps managers command, control, evaluate, and report on operations. Why are the concepts of understand ability and comparability...
-
Carrie Carson: Carrie is a 60-year-old tai chi instructor living in Santa Fe, New Mexico. For many years, she practiced in Hollywood, and because of an acting background, she developed a clientele...
-
Would ocean tides exist if the gravitational pull of the Moon (and the Sun) were somehow equal on all parts of the world? Explain.
-
Candy owns 40% of the stock of Park, Inc., an S corporation. Her stock basis is $25,000, and she loaned $10,000 to the corporation during the year. How much of Park's $100,000 operating loss can...
-
Show how the conglomerate discount leads to an increase in the cost of equity.
-
Cole Company manufactures only two products'a battery charger and a testing machine for automobile engines. An average of 30,000 chargers and 50,000 testers are sold each year. This year, Cole can...
-
Simplify sec (t) - 1 sec (t) to an expression involving a single trig function with no fractions. If needed, enter squared trigonometric expressions using the following notation. Example: Enter sin...
-
From the first quarter 2018 I 0-Q of Groupon, Inc.: Groupon operates online local commerce marketplaces throughout the world that connect merchants to consumers by offering goods and services,...
-
Xylose is a common sugar found in many types of wood, including maple and cherry. Because it is much less prone to cause tooth decay than sucrose, xylose has been used in candy and chewing gum....
-
On catalytic hydrogenation over a platinum catalyst, ribose (Problem 9.57) is converted into ribitol. Is ribitol optically active or inactive?Explain. CH2 Ribitol
-
An absorber is to be designed for a pressure of 75 psia to handle 2,000 lbmol/h of gas at 60F having the following composition: The absorbent is an oil, which can be treated as a pure component...
-
Jack asked Jill to marry him, and she has accepted under one condition: Jack must buy her a new $320,000 Rolls-Royce Phantom. Jack currently has $66,130 that he may invest. He has found a mutual fund...
-
Zhang Industries budgets production of 390 units in June and 400 units in July. Each unit requires 1.5 hours of direct labor. The direct labor rate is $15.80 per hour. The indirect labor rate is...
-
Compute Altoona Company's (a) inventory turnover ratio and (b) number of days' sales in inventory ratio, using the following information. Cost of Goods - $722,000 Beginning Inventory - $53,000 Ending...
-
Fisher's (1936) iris data set provides the measurements in centimeters of the variables sepal length and width and petal length and width, respectively, for 150 flowers (50 flow- ers from each of the...
-
You are offered $120,000 today or $320,000 in 13 years. Assuming that you can earn 13 percent on your money, which should you choose? If you are offered $320,000 in 13 years and you can earn 13...
-
Each of the following situations has an internal control weakness. a. Betty Grable has been your trusted employee for 30 years. She performs all cash-handling and accounting duties. Betty Grable just...
-
Why do bars offer free peanuts?
-
A solution contains Ag + and Hg 2 + ions. The addition of 0.100 L of 1.22 M NaI solution is just enough to precipitate all the ions as AgI and HgI 2 . The total mass of the precipitate is 28.1 g....
-
Oligosaccharides of the type shown in Fig. P24.53 are obtained from the partial hydrolysis of starch amylopectin. What ratio of erythritol to glycerol would be obtained from successive treatment of a...
-
Oligosaccharides of the type shown in Fig. P24.53 are obtained from the partial hydrolysis of starch amylopectin. What ratio of erythritol to glycerol would be obtained from successive treatment of a...
-
Planteose, a carbohydrate isolated from tobacco seeds, can be hydrolyzed in dilute acid to yield one equiva-lent each of D-fructose, D-glucose, and D-galactose. Almond emulsion (an enzyme preparation...
-
ces Exercise 12-9 (Algo) Net Present Value Analysis and Simple Rate of Return [LO12-2, LO12-6] Derrick Iverson is a divisional manager for Holston Company. His annual pay raises are largely...
-
Homework 5 Question 5 of 13 < Current Attempt in Progress On July 1, 2022, Sweet plc made two sales. 1. 2. -/1 E It sold land having a fair value of 908,350 in exchange for a 4-year,...
-
Briefly describe what each of the following terms mean: 1. Annuity 2. Balloon payment 3. Compound interest 4. Discount rate (not Federal Reserve definition) 5. Future value 6. Future value factor 7....

Study smarter with the SolutionInn App


