Show the products of thesereactions: 1) BH3, THF 1) BH3. THF 2) .. NaOH b) 2) ,,
Question:
Show the products of thesereactions:
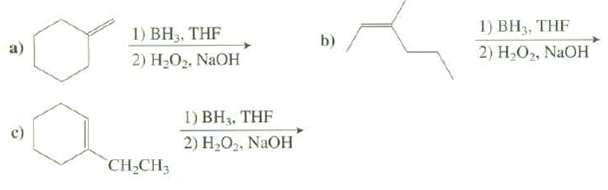
Transcribed Image Text:
1) BH3, THF 1) BH3. THF 2) Н.О. NaOH b) 2) Н,О, NaOH 1) BH3, THF 2) H,O2. NaOH CH-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a OH o b ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
You start driving east for 14 miles, turn left, and drive north for another 10 miles. At the end of driving, what is your straight line distance from your starting point? Round to the nearest tenth...
-
As CEO of Aqua Marine, Carrie Easton knows it is important to control costs and to respond quickly to changes in the highly competitive boat-building industry. When Rose Consulting proposes that Aqua...
-
The number of viewers for network television shows has a direct effect on the amount of advertising dollars that will be generate d. The following data show the number of viewers, in millions, for...
-
Use chloroform to extract acetone from water. Equilibrium data are given in Table 13-6. Find number of equilibrium stages required for a countercurrent cascade if feed is \(1000.0 \mathrm{~kg} /...
-
Trecek Corporation incurs research and development costs of $650,000 in 2017, 30 percent of which relate to development activities subsequent to IAS 36 criteria having been met that indicate an...
-
Discuss the impact of globalization on supply chain management and the challenges it presents to businesses.
-
Jo Herrer started Herrers Bicycle Shop in Tilburg at the end of the 1980s, and the business has thrived in this very green university town in the Netherlands. Jo imports bikes from renowned global...
-
Explain which of the reaction would provide a better synthesis of3-hexanone. . . CH,CH,CH,CH,CH, 3-Hexanone CH,CH,C=CCH CH3 CH,C=CCH,CH,CH3 H,SO4 HgSO, H&SO,
-
This hydroboration reaction forms two products. Show these products and explain which one you expect to be amajor. 1) BH3, THF 2) H,O2. NAOH
-
Felix and Oscar applied for the same credit card from the same bank. The bank checked both of their FICO scores. Felix had an excellent credit rating, and Oscar had a poor credit rating. a. Felix was...
-
If all markets were contestable, there would be no need for regulation. Do you agree with this statement? Provide reasons and justifications for your answer.
-
Why is a time lag an important feature of a perfectly contestable market?
-
Why do regulators face a trade-off between competitiveness and the benefit of economies of scale in imperfectly competitive markets?
-
Why is costless exit as important a feature of a perfectly contestable market as costless entry?
-
Why were Leontiefs findings considered paradoxical? Explain.
-
Describe the process of valuing the entrepreneurs ownership interest in a two-stage investment. Refer to Tables 13.10 and 13.13. Data from table 13.10 Data from table 13.13 Market potential...
-
Estimate a range for the optimal objective value for the following LPs: (a) Minimize z = 5x1 + 2x2 Subject to X1 - x2 3 2x1 + 3x2 5 X1, x2 0 (b) Maximize z = x1 + 5x2 + 3x3 Subject to X1 + 2x2 +...
-
Calculate the mass of sodium acetate that must be added to 500.0 mL of 0.200 M acetic acid to form a pH = 5.00 buffer solution.
-
Substituted pyrroles are often prepared by treatment of a 1, 4-diketone with ammonia. Propose amechanism. R + H20 RCCH2CH2R" R- NH3 R.
-
3, 5-Dimcthylisoxazole is prepared by reaction of 2, 4-pentanedionc with hydroxylamine. Propose amechanism. CH CH3CCH2CCH3 + H2NOH 3,5-Dimethylisoxazole
-
Account for the fact that p-nitro aniline (pKa = 1.0) is less basic than m-nitro aniline (pKa = 2.5) by a factor of 30. Draw resonance structures to support your argument. (The PKa values refer to...
-
Sally is the HIM director at Memorial Hospital and has been asked to compose a record retention policy for the hospital. What should be her first consideration in determining how long paper and...
-
Your firm, which operates in the mining sector, is considering a proposal to build a new processing plant on the site of one of its vacant warehouses. The new building will cost $100 million, while...
-
Suppose you buy an 8% percent annual coupon bond for $1,100. The bond has 10 years to maturity. The interest is paid once a year. Five years from now, the interest rates have increased by 3% and you...

Study smarter with the SolutionInn App


