Show the products you would obtain by reduction of the following esters withLiAlH4: (b) (a)
Question:
Show the products you would obtain by reduction of the following esters withLiAlH4:
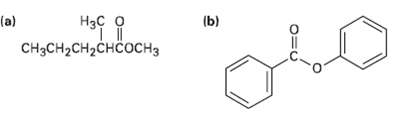
Transcribed Image Text:
(b) Нас о (a) CHясH2CH2снсоснз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (20 reviews)
Lithium aluminum hydride reduces an est...View the full answer

Answered By

Shem Ongek
I am a professional who has the highest levels of self-motivation. Additionally, I am always angled at ensuring that my clients get the best of the quality work possible within the deadline. Additionally, I write high quality business papers, generate quality feedback with more focus being on the accounting analysis. I additionally have helped various students here in the past with their research papers which made them move from the C grade to an A-grade. You can trust me 100% with your work and for sure I will handle your papers as if it were my assignment. That is the kind of professionalism that I swore to operate within. I think when rating the quality of my work, 98% of the students I work for always come back with more work which therefore makes me to be just the right person to handle your paper.
4.80+
174+ Reviews
426+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products you would obtain by acid-catalyzed reaction of Cyclohexanone with ethylamine, CH3CH2NH2, and with diethyl amine, (CH3CH2)2NH.
-
Show the structures of the products you would obtain by hydroboration/oxidation of the following alkenes: H (a) "CH (a) CH3C=CHCH2CH3 (b)
-
Show the products you would except to obtain by claisen condensation of the following esters: (a) (CH3)2CHCH2COEt (b) Ethyl phenyl acetate (c) Ethyl cyclohexylacetate
-
The following data represent the concentration of dissolved organic carbon (mg/L) collected from 20 samples of organic soil. Assume that the population is normally distributed. Complete parts (a)...
-
Why is searching for a job both exhilarating and intimidating? How can you overcome feelings of intimidation?
-
In the 1990 Clean Air Act Amendments, Congress and the EPA rely on the automobile industry to develop a cleaner automobile. At the same time, the government imposes a relatively minor federal tax on...
-
DIVIDENDS Bowles Sporting Inc. is prepared to report the following 2014 income statement (shown in thousands of dollars). Sales $15,200 Operating costs including depreciation 11,900 EBIT $ 3,300...
-
Complex wave vectors in the energy gap find an expression for the imaginary part of the wave vector in the energy gap at the boundary of the first Brillouin zone, in the approximation that led to Eq....
-
Betanet is also experiencing a period of rapid growth. Earnings and dividends are expected to grow at a rate of 15% during the next 2 years, at 11% in the 3rd year, and at a constant rate of 6%...
-
The following information relates to Home Depot, Inc., and Lowes Companies, Inc. for their 2017 and 2016 fiscal years. Required a. Compute the following ratios for the companies 2017 fiscal years...
-
What product would you expect from the reaction of butyrolactone with LiAlH4 with DIBAH? Butyrolactone
-
What ester an what Grignard reagent might you start with to prepare the following alcohols? (b) , H (c) HCH-H2H2,H CH-CH (a)
-
Shaving is a process that most men perform each morning. Assume that the process begins at the bathroom sink with the shaver walking (say, 5 feet) to the cabinet where his shaving supplies are...
-
Swenson Company produced 300 units in year one and sold 260 units in that year. In year two, it produced 260 units and sold 300 units. Total fixed overhead was the same in years one and two. Under...
-
c) Determine the maximum rotational speed such that the fluid will not spill over the container. (and: = 2gh/R) [2 marks] d) The container in Figure 4 now contains coffee (p~1000) which is 7cm deep...
-
FICO credit scores: x = 564,= 743,= 72 (Round your answer to 3 decimal places.) what does z equal
-
Q3: In the section illustrated in Figure (1) the surface 1-4-7 is insulated. The convection heat transfer coefficient at surface 1-2-3 is 28 W/m. 'C. The thermal conductivity of the solid material is...
-
25 of 27 > This test: 96 point(s) possible This question: 3 point(s) possible Submit test Identical twins come from a single egg that split into two embryos, and fraternal twins are from separate...
-
Now that Allisha McKay (see Question 2 above) has compiled the sources and uses of funds information she needs, she is ready to prepare The Fred Proffet Companys Statement of Cash Flows for the Year...
-
What is EBIT/eps analysis? What information does it provide managers?
-
Using the molecular orbital energy ordering for second-row homonuclear diatomic molecules in which the 2p orbitals lie at lower energy than the 2p draw MO energy diagrams and predict the bond order...
-
Rationalize each of the following observations by postulating a structure for the fragment ion(s) and the mechanisms for their formation. The EI mass spectrum of 2-methoxybutane shows a base peak at...
-
Rationalize each of the following observations by postulating a structure for the fragment ion(s) and the mechanisms for their formation. The EI mass spectrum of 2-methoxybutane shows a base peak at...
-
What is the wavelength of light with a wave-number of 1720 cm-1?
-
Production numbers for 2 shifts are shown. The shift supervisor of Shift 2 insists to the production manager that her operators are more productive than the ones on Shift 1. Using a confidence level...
-
In a class, the scores that students got are as shown. What are the 25, 50, 75 and 100th percentiles for the data? 84 84 98 80 89 83 85 56 85 84 84 74 84 81 83 80 45 86 67 79 81 78 76 85 83 77 86 83...
-
Number of points made by Teams A and B are shown. Which statement is true based on running the F-Test Two-Sample for Variances in the Data Analysis pack in Excel? Use a confidence level of 10% to...

Study smarter with the SolutionInn App


