Sketch what you might expect the 1H and 13C NMR spectra of the following compound to look
Question:
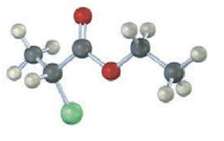
Sketch what you might expect the 1H and 13C NMR spectra of the following compound to look like (yellow-green =Cl):
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The compound has 5 different types of ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The 1H and 13C NMR spectra of compound A, C8H9Br are shown. Propose a structure for A, and assign peaks in the spectra to your structure. TMS O ppm 10 8. 6. Chemical shift (8) TMS 200 180 160 140 120...
-
The proton and 13C NMR spectra of a compound of formula C4H11N are shown here. Determine the structure of this amine, and give peak assignments for all of the protons in the structure. 200 180 160...
-
How could you use 1H and 13C NMR to help you distinguish among the following isomeric compounds of formulaC4H8? CH2-CH2 H2H CH CCH3HCH CH2-CH2 CHH2
-
Suppose, in the New Monetarist model, that there is deficient financial liquidity. If the fiscal authority were to engineer a tax cut, financed by an increase in the quantity of government debt, with...
-
How are franchising and tapered integration similar? How do these strategies differ?
-
1. What is strategic leadership? 2. What would constitute key strategic leadership actions? What are the key elements of a Balanced Scorecard? 3. How has Cheung Yan seen success as a strategic...
-
The degree of psychological tension that is desirable varies with the situation. How can a restaurant manager work to raise or lower the tension to make it appropriate for the situation? AppendixLO1
-
Contrast the buying process for the DOD with buying process used by Target Corporations procurement of laundry detergent from P&G for resale in Target Stores. What aspects of those buying processes...
-
(The jewel box) purchases, jewellery from around the world and sells to local retailers in Canada. Consider the following perpetual system merchandising transactions at the (jewelbox). Use a separate...
-
Information related to property, plant, and equipment; natural resources; and goodwill on December 31, 2021, for H. Dent Company is as follows: land $400,000, building $1,100,000, accumulated...
-
How many absorptions would you expect the following compound to have in its 1H and 13C NMRspectra?
-
How many electronically nonequivalent kinds of protons and how many kinds of carbons are present in the following compound? Don?t forget that cyclohexane rings can ring-flip.
-
Repeat Example 6 with a loan of $6,000 over 5 years. Data from Example 6 A person borrows $3,600 and agrees to repay the loan in monthly installments over 3 years. The agreement is to pay 1% of the...
-
4. (7%) Problem 4: Consider a 570 nm light falling on a single slit of width 1.1 m. Randomized Variables =570 nm w=1.1 um Forbes, David david.forbes@doane.edu @theexpertta.com - tracking id:...
-
(b) The following results are obtained in a double-slit experiment using light from a helium-neon gas laser: Width of 15 fringes = 3.0 cm Separation of slits = 1.5 mm Slit-to-screen distance = 2.5 m...
-
Read the mini-case, Ben and Jerry's Corporate Activism, and answer the following question: What are the pros and cons of Ben and Jerry's political activism when compared to other corporate political...
-
TOPIC : PROBLEMATIZATION - SECOND CURVE THINKING 1. What is second curve thinking?( a More in depth explanation ) 2. What are the implicit assumptions of second curve thinking? ( a More in depth...
-
I think the Power Distance measure in Hofstede's model (Hofstede Insights, n.d.) is particularly interesting.I led divisions in the U.S., New Zealand, and Thailand.Those three countries represented a...
-
In which situations is nonprobability sampling preferred to probability sampling? Comment with examples.
-
What is your opinion of advertising awards, such as the Cannes Lions, that are based solely on creativity? If you were a marketer looking for an agency, would you take these creative awards into...
-
A solution contains 50.0 g of heptane (C 7 H 16 ) and 50.0 g of octane (C 8 H 18 ) at 25 C. The vapor pressures of pure heptane and pure octane at 25 C are 45.8 torr and 10.9 torr, respectively....
-
Predict the chemical shifts of the protons in the following compounds. (a) (b) (c) (d) (e) (f) HC(CH3)3 CH CH CH3 , - CH,CCCH OH CH,CH,-C-OH CH3 CHC-CH Br
-
Draw the NMR spectrum expected from ethanol that has been shaken with a drop of D2O.
-
Propose chemical structures consistent with the following NMR spectra and molecular formulas. In spectrum (a) Explain why the peaks around δ 1.65 and δ 3.75 are not clean...
-
Milano Pizza is a small neighborhood pizzeria that has a small area for in-store dining as well as offering take-out and free home delivery services. The pizzerias owner has determined that the shop...
-
Which of the following statement regarding a post-closing trial balance is not true
-
What are the benefits and potential risks factors for undertaking derivative strategies compared to cash transactions

Study smarter with the SolutionInn App


