Question: For the system shown in Fig. 8.17, compute the power delivered by the pump to the water to pump 50 gal/min of water at 60F
For the system shown in Fig. 8.17, compute the power delivered by the pump to the water to pump 50 gal/min of water at 60°F to the tank. The air in the tank is at 40 psig. Consider the friction loss in the 225-ft-long discharge pipe, but neglect other losses. Then, redesign the system by using a larger pipe size to reduce the energy loss and reduce the power required to no more than 5.0 hp.
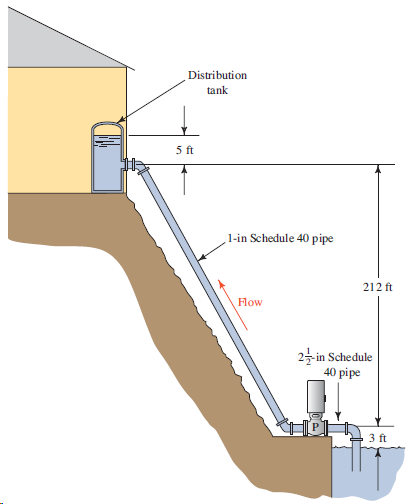
Distribution tank 5 ft 1-in Schedule 40 pipe 212 ft Flow 25-in Schedule 40 pipe 3 ft
Step by Step Solution
3.43 Rating (159 Votes )
There are 3 Steps involved in it
a b Increase the pipe size to 1 in Schedule 40 Results v 788 fts N R 874 10 4 D 895 f 00232 The... View full answer

Get step-by-step solutions from verified subject matter experts


