Arene C has the composition 90.6% carbon and 9.4% hydrogen. Its mass and 1H NMR spectra are
Question:
Arene C has the composition 90.6% carbon and 9.4% hydrogen. Its mass and 1H NMR spectra are shown below.
a.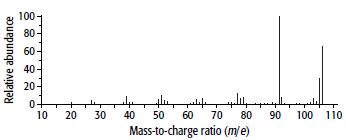
b.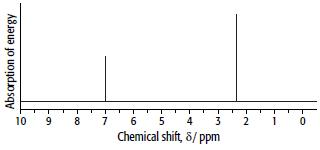
a. Calculate the empirical formula of C.
b. From the mass spectrum, find the molecular mass of C and hence its molecular formula.
c. i. Draw displayed formulae for the possible aromatic isomers of C.
ii. When C is treated with chlorine in the presence of AlCl3 it undergoes electrophilic aromatic substitution. In this reaction one of the hydrogen atoms bonded directly to the benzene ring is replaced with one chlorine atom, and one single aromatic product is formed. Use this evidence and the NMR spectrum of C to decide which isomer is C.
d. Explain the main features of the 1H NMR spectrum of C.
Step by Step Answer:

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris





