A mixture of helium, methane, carbon dioxide, and nitrogen is fed to a perfectly mixed GP system
Question:
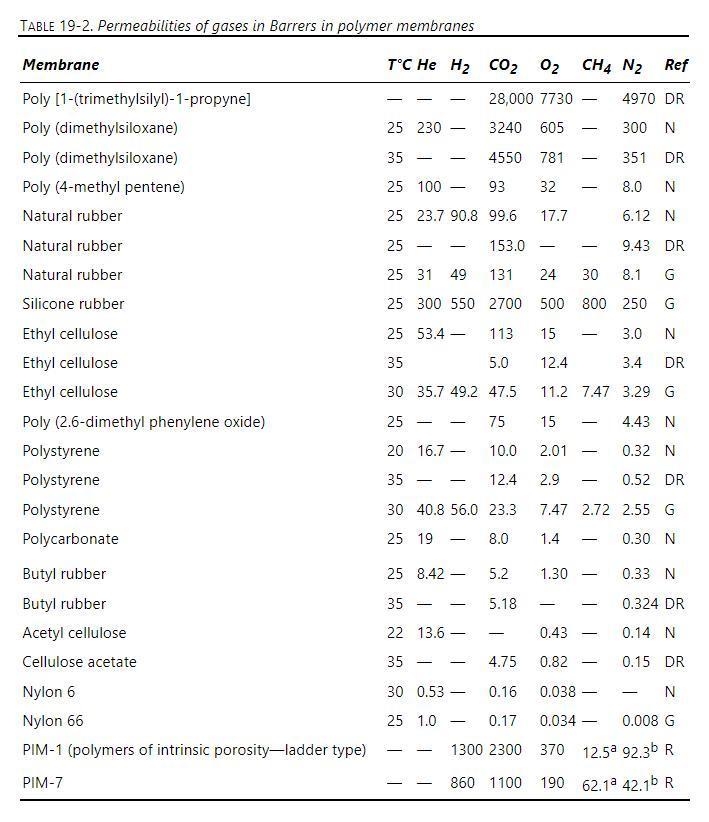
A mixture of helium, methane, carbon dioxide, and nitrogen is fed to a perfectly mixed GP system with a silicone rubber membrane. Membrane properties are given in Table 19-2. Membrane active layer thickness is \(1.0 \mu \mathrm{m}\). Feed pressure is \(12.4 \mathrm{~atm}\) = retentate pressure. Permeate pressure is \(0.92 \mathrm{~atm}\). Feed is \(1.6 \mathrm{~mol} \%\) helium, \(85.5 \mathrm{~mol} \%\) methane, \(2.5 \mathrm{~mol} \%\) carbon dioxide, and \(10.4 \mathrm{~mol} \%\) nitrogen. Feed rate is \(1.25 \mathrm{~m}^{3} / \mathrm{s}\) STP.
a. Find retentate mole fractions, permeate mole fractions, and membrane area (in \(\mathrm{m}^{2}\) ) for \(\theta=0.26\).
b. Find retentate mole fractions, permeate mole fractions, and \(\theta\) if Area \(=10.0 \mathrm{~m}^{2}\).
Step by Step Answer:

Separation Process Engineering Includes Mass Transfer Analysis
ISBN: 9780137468041
5th Edition
Authors: Phillip Wankat





