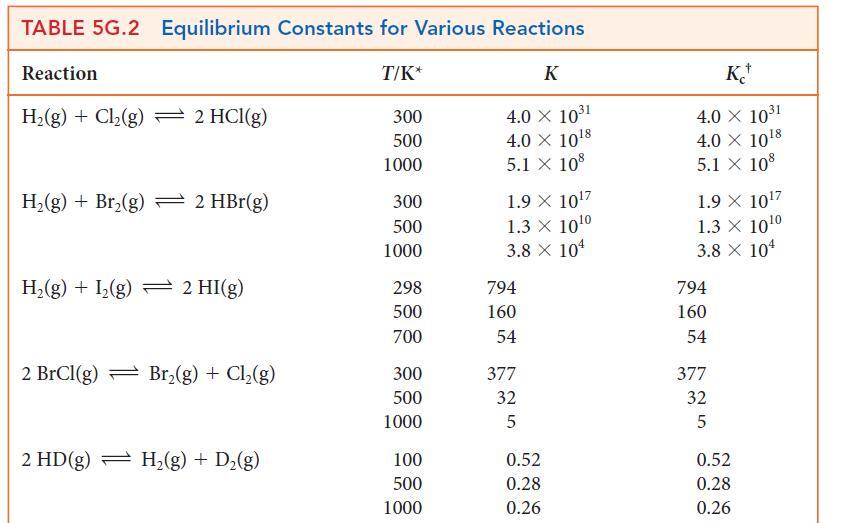
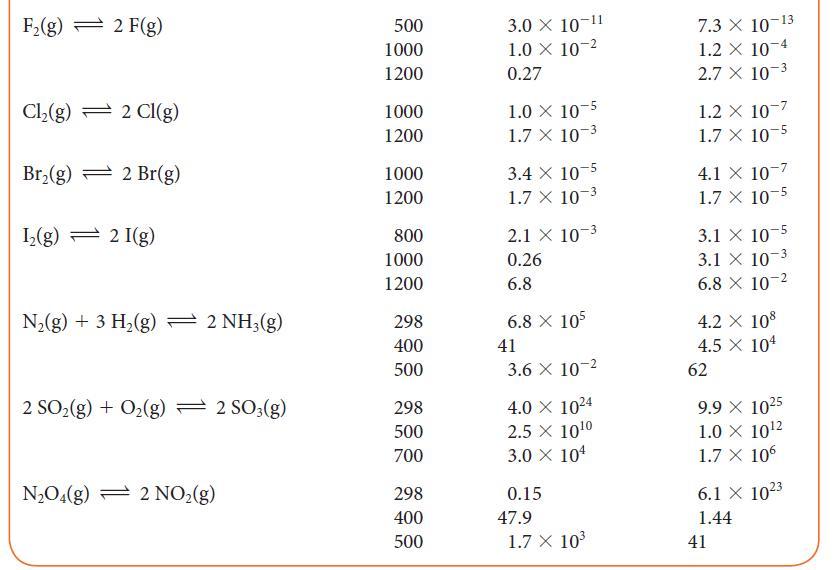
Using data from Table 5G.2 and standard graphing software, determine the standard enthalpy and entropy of the
Question:
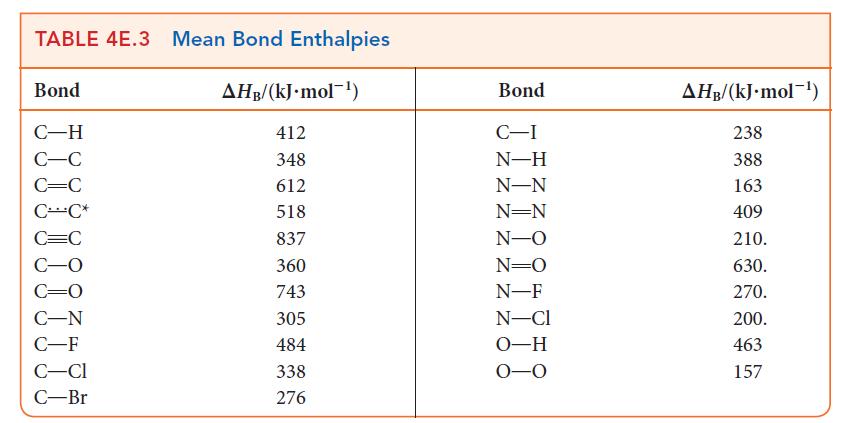
Using data from Table 5G.2 and standard graphing software, determine the standard enthalpy and entropy of the reaction N2O4(g) → 2 NO2(g) and estimate the N—N bond enthalpy in N2O4. How does this value compare with the mean N—N bond enthalpy in Table 4E.3?
Transcribed Image Text:
TABLE 4E.3 Mean Bond Enthalpies AHB/(kJ. mol) Bond C-H C-C C=C CC* C-F C-CI C-Br 412 348 612 518 837 360 743 305 484 338 276 Bond C-I N-H N-N N=N N-O N=O N-F N-Cl O-H 0-0 AHB/(kJ.mol-) 238 388 163 409 210. 630. 270. 200. 463 157
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The graph is generated for In K vs 1T according to this ...View the full answer

Answered By

Atuga Nichasius
I am a Highly skilled Online Tutor has a Bachelor’s Degree in Engineering as well as seven years of experience tutoring students in high school, bachelors and post graduate levels. I have a solid understanding of all learning styles as well as using asynchronous online platforms for tutoring needs. I individualise tutoring for students according to content tutoring needs assessments.
My strengths include good understanding of all teaching methods and learning styles and I am able to convey material to students in an easy to understand manner. I can also assists students with homework questions and test preparation strategies and I am able to help students in math, gre, business , and statistics
I consider myself to have excellent interpersonal and assessment skills with strong teaching presentation verbal and written communication
I love tutoring. I love doing it. I find it intrinsically satisfying to see the light come on in a student's eyes.
My first math lesson that I taught was when I was 5. My neighbor, still in diapers, kept skipping 4 when counting from 1 to 10. I worked with him until he could get all 10 numbers in a row, and match them up with his fingers.
My students drastically improve under my tutelage, generally seeing a two grade level improvement (F to C, C to A, for example), and all of them get a much clearer understanding!
I am committed to helping my students get the top grades no matter the cost. I will take extra hours with you, repeat myself a thousand times if I have to and guide you to the best of my ability until you understand the concept that I'm teaching you.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:
Students also viewed these Sciences questions
-
Which of the following does not define a function? Give its domain and range. A. {(0, 1), (2, 3), (4,8)} B. y = 2x - 6 C. y = x + 2 D. X 0 3 0 6 y 1 2 2 3
-
Listed below is the number of car thefts in a large city over the last week. Calculate the coefficient of skewness using both methods. 13 7 8 3 8 3 12 3
-
The head of a strike anywhere match contains tetraphosphorus trisulfide, P4S3. In an experiment, a student burned this compound in an excess of oxygen and found that it evolved 3651 kJ of heat per...
-
In Exercises 912, use the given conditions to write an equation for each line in point-slope form and general form Passing through (4, -7) and perpendicular to the line whose equation is x - 2y - 3 =...
-
An insulated glass tube and condenser are mounted on a reboiler containing benzene and toluene. The condenser returns liquid reflux so that it runs down the wall of the tube. At one point in the tube...
-
A tutor claimed that getting trained by her will boost scores for an exam. If the before and after scores for 12 students are as shown below, can we say that the tutoring helped using the t-test for...
-
Explain how balance sheets and income statements for manufacturing and merchandising companies differ
-
Arrowhead School District No. 75 is located in Park County, south of Livingston, Montana, and consists of one school, Arrowhead School (School). For the 199798 school year, the School employed about...
-
Dempsey Railroad Co. is about to issue $306,000 of 9-year bonds paying an 12% interest rate, with interest payable semiannually. The discount rate for such securities is 10%. How much can Dempsey...
-
Shibby Shades Inc. manufactures artistic frames for sunglasses. Talia Demarest, controller, is responsible for preparing the companys master budget. In compiling the budget data for 20x1, Demarest...
-
Consider the equilibrium A (g) 2 B (g) + 3 C (g) at 25C. When A is loaded into a cylinder at 10.0 atm and the system is allowed to come to equilibrium, the final pressure is found to be 20.04 atm....
-
The three compounds methylpropene, cis-2-butene, and trans-2-butene are isomers with the formula C 4 H 8 , with G f 5 +58.07, +65.86, and +62.97 kJ mol 1 , respectively. In the presence of a...
-
When translating a foreign operations financial statements into a functional currency, what rate do we apply to monetary assets?
-
We are writing a business plan about expanding Robinson Development Group's business into Mexico. They do residential as well as business development in Virginia and have branched out on the East...
-
Political ideologies can influence trade. Although Russia's constitution lists it as a Federal Democratic State, many news outlets call it a "Fake Democracy". Now that Russia has invaded Ukraine, the...
-
A facultative oxidation pond is to be designed for a community of 5000 people. Summer wastewater flow is 2000 m 3 /d, and the BOD 5 is 180 g/m 3 . Winter flow and BOD 5 values are 6000 m 3 /d and 90...
-
Watch the video "Black Diamond: Managing in a Global Environment" https://youtu.be/lc29Ro9TOKg Describe at least two environmental factors that affect this business and summarize how the managers are...
-
In social media, one size does not fit all. Social media includes traditional platforms such as Facebook and Instagram, but it also includes podcasts, blogs, and video mediums. Each platform and...
-
Can the method of equivalent pipes be used to find a single hypothetical pipe that is equivalent to the pipe system of Problem 3.12.5? If your answer is yes, determine an equivalent pipe. If your...
-
Fill in each blank so that the resulting statement is true. 83 + 103 = ______ .
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) Discuss. OMe . CI H,
-
Deduce the structure of a compound with molecular formula C 5 H 10 O that exhibits the following IR, 1 H NMR, and 13 C NMR spectra. Data from the mass spectrum are also provided. 100 Mass Spec. Data...
-
How do warehouses and distribution centers differ? What is cross-docking and why might a company choose to cross-dock a product? What kinds of products can be delivered electronically? What kinds...
-
Strawberry Inc. has historically been an all-equity firm. The analyst expects EBIT to be $1.5B in perpetuity starting one year from now. The cost of equity for the company is 11.5% and the tax rate...
-
Guzman company received a 60- day, 5 % note for 54,000 dated July 12 from a customer on account. Determine the due date on note. Determine the maturity value of the note and journalize the entry of...

Study smarter with the SolutionInn App


