Gaseous reactant A diffuses through a gas film and reacts on the surface of a solid according
Question:
Gaseous reactant A diffuses through a gas film and reacts on the surface of a solid according to a reversible first-order rate,
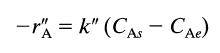
where CA, is the concentration of A in equilibrium with the solid surface. Develop an expression for the rate of reaction of A accounting for both the mass transfer and reaction steps.
Transcribed Image Text:
-ra= k" (CAS- CAe) A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
In order to develop an expression that accounts for both the mass transfer and reaction steps we need to consider the two different rates 1 The mass t...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
In chemical vapor deposition (CVD), a semiconducting or insulating solid material is formed in a reaction between a gaseous species and a species adsorbed on the surface of silicon wafers (disks...
-
The Corporate Average Fuel Economy (CAFE) regulations were put into law by Congress in 1975 to promote the sale of fuel-efficient automobiles and light trucks. The law requires automakers to boost...
-
Discuss the advantages and disadvantages of one of the financial innovations you select. (For example, sweep accounts, junk bond, smart card etcSelect one of them, then explain briefly and afterwards...
-
An article in Sociological Methods & Research (May 2001) analyzed the data presented in the accompanying table. A sample of 262 Kansas pig farmers was classified according to their education level...
-
What problems do analysts often encounter when they try to implement an ERP package?
-
In Exercises 1738, factor each trinomial, or state that the trinomial is prime. 15x 2 - 19x + 6
-
advanced regression techniques of which you should be aware
-
The eight general audit procedures produce evidence about the principal management assertions in financial statements. However, some procedures are useful for producing evidence about certain...
-
Upton umbrellas has a cost of equity of 11.9%, the YTM on the companies bonds is 6.4%, and the tax rate is 21%. The companies bond sell for 103.5% of par. The debt has a book value of 417,000 and...
-
In slurry reactors, pure reactant gas is bubbled through liquid containing suspended catalyst particles. Let us view these kinetics in terms of the film theory, as shown in Fig. P17.3. Thus, to reach...
-
Aqueous A (C AO = 50 mol/m 3 ) with physical properties close to water (p =1000 kg/m 3 , D -9 = m 2 /s) reacts by a second-order reaction (k = m 3 /mol s) as it flows at 10 mm/s through a tubular...
-
1. Consider a thin used-car market. Someone just developed a device that can instantly identify the nearest plum in a used-car lot. The device works only once. The maximum amount that a consumer...
-
What is the logical ending point of a sequential game that starts at position (2,8) with player 1 moving first? Show your work. Player 1 Strategy B Strategy A Strategy A Player 2 Strategy B (3,4)...
-
Problem A-6 Income and Retained Earnings Statements Peanut Corporation is a private corporation using ASPE. At December 31, 2017, an analysis of the accounts and discussions with company officials...
-
8.5 Area Between Curves (dy) Calculus-Calculator Allowed Mastery Check #2 Name: Date: Period: For 1-2, find the area of the region bounded by the following curves. Show the integral set up with...
-
Your company has a travel policy that reimburses employees for the "ordinary and necessary" costs of business travel. Employees often mix a business trip with pleasure by either extending the time at...
-
Simulation A: 1 Diameter 600 mm 2 Focal Length 1800 mm 3 F/D Ratio 3 4 Eyepieces 30 m 5 Barlow? N 6 Celestial Sights M42 - M31 - M51 Simulation B: 1 Diameter 150 mm 2 Focal Length 1800 mm 3 F/D Ratio...
-
The technology underlying hip replacements has changed as these operations have become more popular (over 250,000 in the United States in 2008). Starting in 2003, highly durable ceramic hips were...
-
9.Consider the reaction 3NO2(g)+H2O=2HNO3(aq)+NO(g) where Delta H=-137 kJ.How many kilojoules are released when 92.3g of NO2 reacts?
-
Go to the five LearnChemE screencasts link for Chapter 5 (http://www.umich.edu/~elements/6e/05chap/learn-cheme-videos.html). 1. In the screencast of the PBR with pressure drop, is there a problem...
-
How would you modify Table 6-2 for a. A constant-volume gas-phase reaction, and b. A variable-volume gas-phase reaction? Table 6-2 1. Mole balances: BR dNAdt=rAV dNBdt=rBV dNCdt=rCV dNDdt=rDV PFR PBR...
-
(a) Without referring back, make a list of the most important items you learned in this chapter. (b) Overall, what do you believe were the three major purposes of the chapter?
-
Long-term liabilities are shown in two places in the business firm's balance sheet depending upon when the long-term liabilities are scheduled for payment. True False
-
Julio is single with 1 withholding allowance. He earned $1,025.00 during the most recent semimonthly pay period. He needs to decide between contributing 3% and $30 to his 401(k) plan. If he chooses...
-
Acquirer firm plans to launch a takeover of Target firm. The manager of Acquirer indicates that the deal will increase the free cash flow of the combined business by $13.6m per year forever. The beta...

Study smarter with the SolutionInn App


