Name this alkane: CH-CH-CH-CH-CH-CH-CH-CH3 CH I CH3 CH3 CH3
Question:
Name this alkane: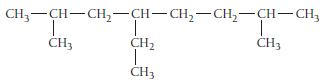
Transcribed Image Text:
CH₂-CH-CH₂-CH-CH₂-CH₂-CH-CH3 CH₂ I CH3 CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
This compound has eight carbon atoms in its longest continuous chain CH3...View the full answer

Answered By

FREDRICK MUSYOKI
Professional Qualities:
Solution-oriented.
Self-motivated.
Excellent problem-solving and critical thinking skills.
Good organization, time management and prioritization.
Efficient troubleshooting abilities.
Tutoring Qualities:
I appreciate students as individuals.
I am used to tailoring resources for individual needs.
I can integrate IT into student's lessons.
I am good at explaining concepts.
I am able to help students progress.
I have a wide curriculum knowledge.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
a. An alkane with molecular formula C5H12 forms only one monochlorinated product when heated with Cl2 Give the systematic name of this alkane. b. An alkane with molecular formula forms C7H16 seven...
-
a. Ethene, C 2 H 4 , is the starting material for making plastic carrier bags. (i) Name the type of chemical change taking place in the diagram above. (ii) Name the product formed by this reaction....
-
Agree or disagree The first logical fallacy I chose is Plain Folks. One of the main examples of this that I have seen is during the beginning months of the Covid-19 pandemic, many celebrities would...
-
Determine the radius of convergence of the following power series. Then test the endpoints to determine the interval of convergence. 2k (x - 3)k k
-
Andy Corporations management took the following actions, which went into effect on January 2, 2010. Each action involved an application of present value. a. Andy Corporation enters into a purchase...
-
What is outplacement, and how does it usually work?
-
What are the main benefits to a business of global sourcing? LO.1
-
Slopes Inc. manufactures and sells snowboards. Slopes manufacture a single model, the Pipex. In the summer of 2012, Slopess accountant gathered the following data to prepare budgets for 2013. These...
-
The pension plan was amended last year, creating a prior service cost of $200 million. Service cost and interest cost for the year were $50 million and $24 million, respectively. At the end of the...
-
What is the correct formula for the alkane (noncyclical) containing eight carbon atoms? a) C 8 H 16 b) C 8 H 18 c) C 8 H 14 d) C 8 H 8
-
Which molecule exhibits optical isomerism? H (a) H-C-cl Br (b) Br H H | | C-C-H cl H H H | | (c) Br C-c-c J-H CIH H (d) Br H 1 C-H . ICIH C-C-Cl H
-
The debt-to-equity ratio of Allied Domecq plc (spirits group) was 2.1 mid 2004. State your views.
-
The employee.class.php file contains an abstract, base class named Employee . One of the attributes of the class is an object of Person . This demonstrates one of the three relationship types among...
-
A direct shear test is performed on a saturated specimen of loose sand. A normal stress equal to 100 kPa is applied and a maximum shear stress of 75 kPa is measured in the shear test. Determine the...
-
How does budgeting help managers? Budgeting helps managers determine if their goals are ethical and achievable. Budgeting helps managers determine if their goals are reasonable and achievable....
-
If the 230-lb block is released from rest when the spring is unstretched, determine the velocity of the block after it has descended 5ft . The drum has a weight of 70lb and a radius of gyration of...
-
A B C D E F 1 Frequency : Monthly 2 Loan Amount: 150000.00 3 Interest Rate: 7.25% 4 Term(years): 30.00 5 No. of payments in a year: 12 time(s) 6 Periodic Rate: 0.60% =C3/C5 7 Total number of payment:...
-
Assume that Lundgren Corporation has a subsidiary company based in Japan? Yen Assets ........................... 410,000,000 Liabilities ....................... 115,000,000 Stockholders' equity:...
-
Explain briefly what is meant by electronic data interchange (EDI). How does EDI affect a companys audit trail?
-
An electric field is given by E (Vector) = E 0 (y/a)k, where E 0 and a are constants. Find the flux through the square in the x-y plane bounded by the points (0, 0), (0, a), (a, a), (a, 0).
-
A nonconducting square plate 75 cm on a side carries a uniform surface charge density. The electric field strength 1 cm from the plate, not near an edge, is 45 kN/C. Whats the approximate field...
-
A 250-nC point charge is placed at the center of an uncharged spherical conducting shell 20 cm in radius. Find (a) the surface charge density on the outer surface of the shell (b) the electric field...
-
Comfort Golf Products is considering whether to upgrade its equipment Managers are considering two options. Equipment manufactured by Stenback Inc. costs $1,000,000 and will last five years and have...
-
Weaver Corporation had the following stock issued and outstanding at January 1, Year 1: 71,000 shares of $10 par common stock. 8,500 shares of $60 par, 6 percent, noncumulative preferred stock. On...
-
Read the following case and then answer questions On 1 January 2016 a company purchased a machine at a cost of $3,000. Its useful life is estimated to be 10 years and then it has a residual value of...

Study smarter with the SolutionInn App


