Name this alkane: HO HD HD-HO-H-HD - HD
Question:
Name this alkane: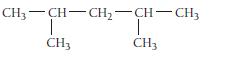
Transcribed Image Text:
HO HD HD-HO-H₂-HD - HD
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
1 The longest continuous carbon chain has five atoms Therefore th...View the full answer

Answered By

Hassan Ali
I am an electrical engineer with Master in Management (Engineering). I have been teaching for more than 10years and still helping a a lot of students online and in person. In addition to that, I not only have theoretical experience but also have practical experience by working on different managerial positions in different companies. Now I am running my own company successfully which I launched in 2019. I can provide complete guidance in the following fields. System engineering management, research and lab reports, power transmission, utilisation and distribution, generators and motors, organizational behaviour, essay writing, general management, digital system design, control system, business and leadership.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
a. An alkane with molecular formula C5H12 forms only one monochlorinated product when heated with Cl2 Give the systematic name of this alkane. b. An alkane with molecular formula forms C7H16 seven...
-
a. Ethene, C 2 H 4 , is the starting material for making plastic carrier bags. (i) Name the type of chemical change taking place in the diagram above. (ii) Name the product formed by this reaction....
-
Agree or disagree The first logical fallacy I chose is Plain Folks. One of the main examples of this that I have seen is during the beginning months of the Covid-19 pandemic, many celebrities would...
-
Evaluate the limit of the sequence or state that it does not exist. an || 1 + 32n n
-
JetGreen Airways instituted a frequent flyer program in which passengers accumulate points toward a free flight based on the number of miles they fly on the airline. One point was awarded for each...
-
Find the t table value that would be used to construct the confidence interval for the slope coefficient in a regression analysis for each of the following situations: a. For 95% confidence, based on...
-
The National Trust for Historic Preservation is a private, nonprofit membership organization dedicated to saving historic places and revitalizing America's communities. Having been chartered by...
-
Alard Company produces blenders and coffee makers. During the past year, the company pro-duced and sold 65,000 blenders and 75,000 coffee makers. Fixed costs for Alard totaled $340,000,of which...
-
**#6.) In right AJKL below, a) Write the trigonometric ratio to find the m/]J and solve for m/J. Round to the nearest tenth. K 22 L b) *Write the trigonometric ratio to find the mZK and solve for...
-
Arrange the compounds from least oxidized to most oxidized. (i) CH3 C CH3 (iii) CH3-CH-CH3 (a) iii
-
The formulas of three noncyclic hydrocarbons are listed below. Which of these is an alkene? (a) C 4 H 10 (b) C 3 H 4 (c) C 5 H 10
-
If h = 5 m, determine the maximum tension developed in the chain and its length. The chain has a mass per unit length of 8kg/m. 50 m h = 5 m
-
If packing costs are directly proportional to the volume of packing, show that packing costs go through a minimum as L/D increases.
-
Biorefineries producing ethanol by fermentation have several distillation columns to separate the ethanol from the water. The first column, the beer still, is a stripping column that takes the dilute...
-
Consider the production function given in Problem 3. Assume that \(N\) is constant and equal to 1 . Note that if \(z=x^{a}\), then \(g_{z} \approx a g_{x}\), where \(g_{z}\) and \(g_{x}\) are the...
-
Which of the following is not a component of net pension expense? a. Interest cost b. Expected return on plan assets c. Benefits paid to retirees d. Amortization of actuarial gains or losses
-
When the value of pension plan assets is greater than the projected benefit obligation, a. the difference is added to pension expense. b. the difference is reported as deferred pension cost. c. the...
-
This excerpt comes from an article titled Eagle Eyes High-Coupon Callable Corporates in the January 20, 1992, issue of BondWeek, p. 7: If the bond market rallies further, Eagle Asset Management may...
-
A. Select a recent issue (paper or online) of Report on Business Magazine, Canadian Business Magazine (online only), Bloomberg Businessweek, Fast Company, The Economist, or another business magazine....
-
An infinitely long rod of radius R carries a uniform volume charge density . Show that the electric field strengths outside and inside the rod are given, respectively, by E = R 2 /2 0 r and E = r/2...
-
A long, solid rod 4.5 cm in radius carries a uniform volume charge density. If the electric field strength at the surface of the rod (not near either end) is 16 kN/C, whats the volume charge density?
-
If you painted positive charge on the floor, what surface charge density would be necessary to suspend a 15C, 5.0-g particle above the floor?
-
T4 information slips and T4 summary forms must be submitted by: Multiple Choice The final day of February of the following year A convenient date determined by the employer 30 days after the year-end...
-
During 2021/22 Tax Year, Selma Car (aged 63), received a superannuation income stream benefit of $57,200 from her superannuation funds. As her superannuation was derived from an untaxed element...
-
COMPREHENSIVE PROBLEM 2 - The following ledger accounts appear below, in this order: Cash, Accounts Receivable, Inventory, Estimated Returns Inventory, Prepaid Insurance, ...

Study smarter with the SolutionInn App


