Calculate r G and the equilibrium constant for the following reactions. (a) 2 Fe+ (aq) +
Question:
Calculate ΔrG° and the equilibrium constant for the following reactions.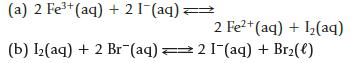
Transcribed Image Text:
(a) 2 Fe³+ (aq) + 21 (aq): (b) 1₂(aq) + 2 Br(aq) = 2 Fe²+ (aq) + ₂(aq) 21-(aq) + Br₂(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To calculate rG standard Gibbs free energy change and the equilibrium constant K for these reactions ...View the full answer

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Calculate the equilibrium constant for the acid-base reactions between the following pairs of reactants. a. HCl + H2O b. CH3COOH + H2O c. CH3NH2 + H2O CH3NH3 + H20
-
The molecule methylamine (CH3NH2) can act as a monodentate ligand. The following are equilibrium reactions and the thermochemical data at 298 K for reactions of methylamine and en with Cd2+ (aq); (a)...
-
Calculate the standard free-energy change and the equilibrium constant Kp for the following reaction at 25oC. See Appendix C for data. CO(g) + 2H2(g) =CH,OH(g)
-
On 1 January 2022, ABC Company issued 10,000 shares of common stock for $100,000. On 31 December 2022, the company declared and paid dividends of $10,000. Calculate the earnings per share and the...
-
The following data were taken from the records of Nomad Company for March 2008: Administrative expenses ............... $ 58,000 Cost of goods sold (at standard) ............ 885,500 Direct materials...
-
Its easier to learn accounting if you avoid real-world examples. Do you agree? Explain.
-
What are the advantages and disadvantages to the opt-in versus the opt-out approaches to collecting personal information from the viewpoint of the customer? From the viewpoint of the organization...
-
Oil Products Company purchases an oil tanker depot on January 1, 2009, at a cost of $600,000. Oil Products expects to operate the depot for 10 years, at which time it is legally required to dismantle...
-
If you were in charge of deciding how healthcare providers would get paid for their services, what steps would you take to restructure their payments? It's not about which method you would choose,...
-
One half-cell in a voltaic cell is constructed from an iron electrode in an Fe(NO 3 ) 2 solution of unknown concentration. The other half-cell is a standard hydrogen electrode. A potential of 0.49 V...
-
One half-cell in a voltaic cell is constructed from a silver wire electrode in a AgNO 3 solution of unknown concentration. The other half-cell consists of a zinc electrode in a 1.0 M solution of...
-
UAA is a stop codon. Why does the UAA sequence in mRNA in Problem 18 not cause protein synthesis to stop?
-
SDN company has two divisions. Division A makes a component X, which is transferred to the Division B. Component X is further processed by the Division B and is sold to customers at a price of $200...
-
1.For each of the following activities, identify whether the main role of accounting is (1) problem solving, (2) scorekeeping, or (3) attention directing. Item: a.Analyzing the impact of introducing...
-
How do contemporary theories of work-life balance and employee well-being align with organizational performance metrics and long-term business sustainability ?
-
to the transactions above. 2. (25p.) Ali buys pencils for $2 each, and sells them in the Taxim Square for $5 each. He sells 20 pencils in a day. Total sales decrease 2 units for every $ price...
-
Line 9 10 11 1. Create the table that contains the following information for the last available quarter. Please note that sing the data for previous quarters will produce grade zero for this part of...
-
ATT's dividend history from 2006-2011 is shown below. a.) Calculate the 1-, 3-, and 5-year average annual rates of growth in ATT's annual dividend. b.) Has ATT's dividend growth been accelerating or...
-
Read the case study Richter: Information Technology at Hungarys Largest Pharma and answer the following question: How does the organization ensure the accuracy of the data it stores?
-
A roofing tile falls from rest off the roof of a building. An observer from across the street notices that it takes 0.43 s for the tile to pass between two windowsills that are 2.5 m apart. How far...
-
You are standing at the top of a deep, vertical cave and want to determine the depth of the cave. Unfortunately, all you have is a rock and a stopwatch. You drop the rock into the cave and measure...
-
Your friend is an environmentalist who is living in a tree for the summer. You are helping provide her with food, and you do so by throwing small packages up to her tree house. If her tree house is...
-
Explain how investors react to changes in a company's dividend payout policy. Discuss what a firm's payout policy indicates about the financial status of a company. Discuss how taxes impact an...
-
Hi, please help with this question. I was able to answer part a but have gotten stuck on B and C. Thank you (Efficiency analysis) The Brenmar Sales Company had a gross profit margin(gross...
-
John currently has a portfolio of shares giving a return of 20% with a risk of 10%. He is considering a new investment which gives a return of 20% with a risk of 12%. The coefficient of correlation...

Study smarter with the SolutionInn App


