What volume of 0.125 M oxalic acid, H 2 C 2 O 4 , is required to
Question:
What volume of 0.125 M oxalic acid, H2C2O4, is required to react with 35.2 mL of 0.546 M NaOH?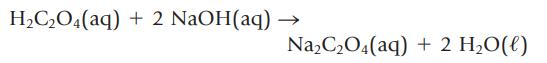
Transcribed Image Text:
H₂C₂O4(aq) + 2 NaOH(aq) Na₂C₂O4(aq) + 2 H₂O(l)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To determine the volume of 0125 M oxalic acid H2C2O4 required to react with 352 mL of 0546 M N...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Marnie wants to save up $250,000 to pay cash for a home purchase 15 years from now. If her investment can carn 6.1% compounded monthly and she intends to grow each payment by 0.25%, what will be her...
-
1. What mass of H2 should be produced by the reaction of Al with 75.0 mL of 2.95M HCl? 2Al(s) + 6HCl(aq) 2AlCl3(aq) + 3H2 (g). ln the lab, 0.15g H2 was collected. What is the % yield of the...
-
For Reaction 1-7, how many milliliters of 0.165 0 M KMnO4 are needed to react with 108.0 mL of 0.1650 M oxalic acid? How many milliliters of 0.1650 M oxalic acid are required to react with 108.0 mL...
-
Landry State University, a public university located in Louisiana, has a June 30 fiscal year. On July 20 of the current year, Landry State University receives $2,125,000 in payments from the U.S....
-
Describe the concept of Six Sigma quality. Why is such a high quality level important?
-
Collars A and B are connected by a 1-m-long wire and can slide freely on frictionless rods. If a force (P= 680 N)j is applied at A, determine (a) The tension in the wire when y = 300 mm, (b) The...
-
Trading volume on the New York Stock Exchange is heaviest during the first half hour (early morning) and last half hour (late afternoon) of the trading day. The early morning trading volumes...
-
As of the end of June, the job cost sheets at Racing Wheels, Inc., show the following total costs accumulated on three custom jobs. Job 102 was started in production in May and the following costs...
-
thank you Listed here are data for five companies. These data are for the companies 2019 fiscal years. The market price per share is the closing price of the companies' stock the day after they...
-
What volume of 0.812 M HCl, in milliliters, is required to titrate 1.45 g of NaOH to the equivalence point? NaOH(aq) + HCl(aq) HO(l) + NaCl(aq)
-
What volume of 0.750 M Pb(NO 3 ) 2 , in milliliters, is required to react completely with 1.00 L of 2.25 M NaCl solution? The balanced equation is Pb(NO3)2(aq) + 2 NaCl(aq) PbCl(s) + 2 NaNO3(aq)
-
Repeat Exercise 6 using the method of False Position. a. ex + 2x + 2 cos x 6 = 0 for 1 x 2 b. ln(x 1) + cos(x 1) = 0 for 1.3 x 2 c. 2x cos 2x (x 2)2 = 0 for 2 x 3 and 3 x 4 d. (x 2)2 ...
-
Directions: Put your feet in the shoes of the business owner and suggest specific ways on how a business can gain profit and how it can be avoid loss Ways to Gain Profit 1. 2. 3. 4. 5. 1. 2. 3. 4. 5....
-
Define business intelligence Briefly discuss how your organisation can use business intelligence to improve decision-making Please see below rubric as guidance. Kindly list references in APA 7th...
-
Mr Santos apply for college educational plan for his 3 children .The 3 children ages are 6 yrs old , 3 yrs old and 1 yr old. The fund will be set-up the deposit of a fixed sum on the child's current...
-
Recovery Centers of America needs to acquire new vehicles that will cost $2.5 million across its six state service area. It plans to use the vehicles for three years, at which time new vehicles will...
-
The following program is supposed to allow the user to enter a list of numbers, then print them out formatted with 2 decimal positions. However, there are 3 errors. Indicate the line number of each...
-
Heather Hutchison is a realtor. She organized the business as a corporation on March 10, 2008. The business received $50,000 cash from Hutchison and issued common stock. Consider the following facts...
-
Consider the setup in Problem 16. Show that the relative speed of the ball and the point of contact on the stick is the same before and immediately after the collision. (This result is analogous to...
-
The Chalk Point, Maryland, generating station supplies electrical power to the Washington, D.C., area. Units 1 and 2 have a gross generating capacity of 710. MW (megawatt). The steam pressure is 25 ...
-
An electrical motor is used to operate a Carnot refrigerator with an interior temperature of 0.00C. Liquid water at 0.00C is placed into the refrigerator and transformed to ice at 0.00C. If the room...
-
Below are two hypothetical compounds. a) Which compound would you expect to hold greater promise as a potential antihistamine? Explain your choice. b) Do you expect the compound you chose (in part a)...
-
Phone Corporation owns 80 percent of Smart Company's common stock, acquired at underlying book value on January 1, 20x4. At the acquisition date, the book values and fair values of Smart's assets and...
-
The payment of principal in pay-troughs is used to create a sinking fund whose value will equal the principal due on each subsequent tranche at maturity pay the residual class retire each tranche in...
-
which tow states tax dividends and interest income but do not tax earned income? New Hampshire and Kentucky Kentucky and Conneticut connecticut and Tennessee New Hampshire and Tennessee
Liberate Sports Tracking Devices For Traveling Outdoor Fitness Backup - ISBN: B0CGQTM4GL - Free Book

Study smarter with the SolutionInn App


