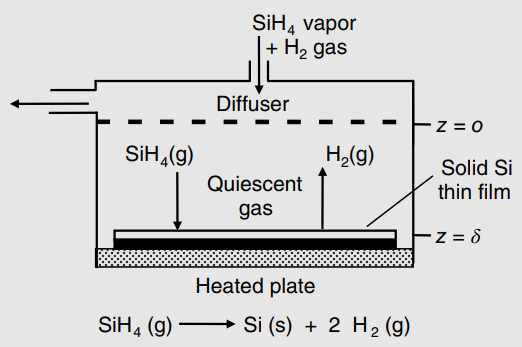
The chemical vapor deposition of silane vapor (SiH 4 ) forms a thin film of solid silicon
Question:
composition of 20 mole% silane. The Lennard€“Jones para meters for SiH4 are σA = 4.08A and ϵA/k = 207.6 K.
a. Recall from example 1, Chapter 25 that the steady-state molecular diffusion flux of silane vapor (species A) down to the silicon wafer is 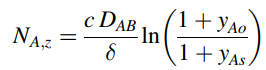
where yAo is the mole fraction of silane in the bulk gas, and yAs is the mole fraction of silane vapor at the surface of the wafer. Assume that surface reaction is instantaneous, so that the rate of silane decomposition to solid silicon is controlled by the molecular diffusion of silane to the silicon surface (e.g., YAs ‰ˆ 0). Estimate the rate of Si film formation in units of microns (µm) of Si solid film thickness per minute. The density of crystalline silicon is 2.32 g/cm3.
b. Now consider that the surface reaction is not instantaneous, so that YAs > 0. The simplified rate equation is given by
NA|z=δ = + ks CAs
where the (+) sign indicates that the direction of flux for species A is in the same direction as increasing z. At 900 K, ks = 1.25 cm/s. using data provided by Middleman and Hochberg (1993).* Develop a revised model for the flux NA, which does not contain the y term, using the approximation ln( 1 + y) =̇ y. From this model, estimate the rate of Si film formation, and then compare the result to part (a) above. Comment on the importance of diffusion versus surface reaction in determining the silicon thin film formation rate.
Figure 25.5
Step by Step Answer:

Fundamentals Of Momentum Heat And Mass Transfer
ISBN: 9781118947463
6th Edition
Authors: James Welty, Gregory L. Rorrer, David G. Foster





