Use the information given here, data from Appendix D, and equation (7.22) to calculate the standard enthalpy
Question:
Use the information given here, data from Appendix D, and equation (7.22) to calculate the standard enthalpy of formation per mole of ZnS(s).
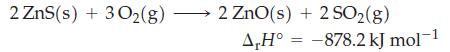
Eq.7.22
![A.H = [cx AHC + dx AHD +...] [ax AHA + bx AHB + .] (7.22) weighted sum of AcH values for the products](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1699/6/0/4/689654de8d1674691699604684867.jpg)
Transcribed Image Text:
2 ZnS(s) + 3O₂(g) 2 ZnO(s) + 2 SO₂(g) A,H° -878.2 kJ mol-1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
2ZnSs 302g 2ZnO2SO2g AH rxn...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Calculate the standard enthalpy of formation of NOCI (g) from the enthalpy of formation of NO given in Table 2.5, together with the following information: 2 NOCl (g) 2 NO (g) + Clz (g) 1 Ho = + Uo=...
-
Calculate the standard enthalpy of formation for diamond, given that C(graphite) + O2(g) CO2(g) Afr =-393.5 kJ/mol C(diamond) + O2(g) CO2(g) AF1 395.4 kJ/mol
-
The average energy output of a good grade of coal is 2.6 10 7 kJ/ton. Fission of 1 mol of 235 U releases 2.1 10 10 kJ. Find the number of tons of coal needed to produce the same energy as 1 lb of...
-
You have obtained the following share prices from Yahoo Finance. Company 1 is listed on the London Stock Exchange. You want to estimate the beta value for Company 1. You run a regression of the share...
-
The following calendar year-end information is taken from the December 31, 2009, adjusted trial balance and other records of Gucci Company. Advertising expense . . . . . . . . . . . . . . . . . . . ....
-
What is the difference between a conventional mortgage and a high ratio mortgage? What is a vendor take-back mortgage? How does it work?
-
CAPITAL BUDGETING CRITERIA: MUTUALLY EXCLUSIVE PROJECTS A firm with a WACC of 10% is considering the following mutually exclusive projects: 0 2 3 $225 $49 $225 $50 $55 $50 $55 $300 $55 $300 2$400...
-
Finer Company uses a sales journal, a purchases journal, a cash receipts journal, a cash disbursements journal, and a general journal. The following transactions occur in the month of May. May 2 Sold...
-
Die #1 Rol Possible Outcome 1 2 3 4 5 6 Die #2 1 1 1 1 9.00 1st Roll "Coming out of Odds Winning (7 or 11) Cras 2,3 or 12) Points 2 3 4 5 Pass Line Bel - Pays Even Money lie $1 staket on winget $1 +...
-
Q1: (50 Mark) Given the following noise data, calculate LAeq, L10, L50,L90, Nc and Lap: Time(sec) SPL(dBA) 10 70 20 50 30 65 40 60 50 55 60 65 70 60 80 55 90 70 100 50
-
Use the data in Figure 7-18 and information to establish possible relationships between the molecular structure of the hydrocarbons and their standard enthalpies of formation. Figure 7-18 Positive...
-
The standard enthalpy of fermentation of glucose to ethanol is Use the standard enthalpy of combustion for glucose to calculate the enthalpy of combustion for ethanol. C6H12O6(s)- 2 CH3CHOH(1) + 2...
-
When common stock is sold by a corporation a journal entry is prepared which includes a debit to cash and a credit to the common stock account. If the debit to cash is greater than the credit to the...
-
33. If the equipment in the previous question had sold for $15,000, the correct entry would be: a. Cash debit $15,000. Gain credit $3,000. $12,000 Equipment credit b. Cash debit $15,000. Debit a loss...
-
The banks play a central role in financial intermediation in New Zealand. 1.What is financial intermediation? Who performs it? and why is it important? 2.What is Qualitative Asset transformation...
-
Consider the following information attributed to the material management department Budgeted usage of materials - handling labor - hours 3,700 Budgeted cost pools: Fixed costs $166,500 Variable costs...
-
5) A frictionless rod of length L rotates counterclockwise in the with constant angular speed w at an angle a to the z axis. A bead of mass m, free to slide on the rod, leaves the origin with initial...
-
1) Louisa is a corn farmer in Illinois. She anticipates a harvest in August of 3 million bushels of yellow corn. Today is May. Louise plans to hedge her sale of corn in August using corn futures...
-
How are carbide tools manufactured?
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
State how each of the following items is reflected in the financial statements. (a) Change from FIFO to LIFO method for inventory valuation purposes. (b) Charge for failure to record depreciation in...
-
Discuss briefly the three approaches that have been suggested for reporting changes in accounting principles.
-
Identify and describe the approach the FASB requires for reporting changes in accounting principles.
-
Shown below is activity for one of the products of Denver Office Equipment: January 1 balance, 620 units @ $55 per unit $34,100 Purchases: January 10: 620 units @ $60 per unit January 20: 1,240 units...
-
QUESTION 1 ( 2 0 Marks ) A company manufactures and sells a single product. Budgeted data per unit of the product is: \ table [ [ , R 1 . 1 Prepare statements of Comprehensive income for both periods...
-
12. Accrual for product warranty liability could result in Future Future Taxable Amounts Deductible Amounts a. Yes Yes b. Yes No c. No Yes d. No No 13. Subscriptions received in advance could result...

Study smarter with the SolutionInn App


