Flash calculations are simpler for binary systems than for the general multicomponent case because the equilibrium compositions
Question:
Flash calculations are simpler for binary systems than for the general multicomponent case because the equilibrium compositions for a binary are independent of the overall composition. Show that, for a binary system in VLE
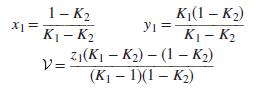
Transcribed Image Text:
1- K2 K1- K2 z1(KI – K2) – (1 – K2) K(1 – K2) Yi = K1- K2 ソ= (K1 – 1)(1 – K2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
For a binary system in VLE the equilibrium compositions are determined by the equilibrium relationsh...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Question Posted:
Students also viewed these Engineering questions
-
A system for separating a binary mixture by extractive distillation, followed by ordinary distillation for recovery and recycle of the solvent, is shown in Figure. Are the design variables shown...
-
Show that By comparing the integrand to a simpler function 10
-
Show that each system has an equilibrium point at the origin. Compute the Jacobian, then discuss the type and stability of the equilibrium point. Find and describe other equilibria if they exist. x'...
-
Miami Tropical stock has a beta of 1.25. The risk-free rate of return is 4.34 percent and the market risk premium is 9.02 percent. What is the expected rate of return on this stock? 10.23 percent...
-
Make an attempt to calculate the present value of your future income.
-
Analysis of the accounts of Jonimatt State College for the fiscal year ended June 30, 20X7, provided the following information: Required Additional Information 1. Earnings of the endowments included...
-
What are the characteristics of a liquid asset? Give examples of some liquid assets. AppendixLO1
-
Use Appendix B.5 to locate the value of t under the following conditions. a. The sample size is 15 and the level of confidence is 95%. b. The sample size is 24 and the level of confidence is 98%. c....
-
Presidio, Inc.. produces one model of mountain bike. Partial information for the company follows: Required: 1. Complete Presidio's cost data table 2. Calculate Presidio's contribution margin ratio...
-
The number of nonconforming switches in samples of size 150 is shown in Table 6E.2. Construct a fraction nonconforming control chart for these data. Does the process appear to be in control? If not,...
-
The excess Gibbs energy for binary systems consisting of liquids not too dissimilar in chemical nature is represented to a reasonable approximation by the equation: where A is a function of...
-
The following is a rule of thumb: For a binary system in VLE at low pressure, the equilibrium vapor-phase mole fraction y 1 corresponding to an equimolar liquid mixture is approximately where P sat i...
-
Evaluate the sum that converges. Identify any that diverge. Write 0.333 . . . as an infinite geometric series. Find the sum.
-
Gordon Rivers, the city manager of Saratoga, Florida, pitched the proposed design schedule back at Jay Andrews. Jay Andrews is the project manager for Major Design Corporation (MDC). The city of...
-
Use the data from SE3-8 to prepare the closing entries for The Decade Company. Close the temporary accounts straight to retained earnings. The balance of \(\$ 8,500\) in the retained earnings account...
-
Draw a Keynesian cross diagram to show the effects of a rise in autonomous expenditure on an economy operating below full employment output.
-
Governments in many countries are acutely aware of the environmental problems that vehicle emissions can have. Many car manufacturers are exploring the production of electric vehicles, but production...
-
Draw a simple diagram of John Woodens pyramid of success. You can find it at the official Wooden website www.coachwooden.com/index2.html.
-
Condensed financial data of Granger Inc. follow. Additional information: 1. New plant assets costing $100,000 were purchased for cash during the year. 2. Old plant assets having an original cost of...
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
As suggested by Fig. 3.1, the slope of the sublimation curve at the triple point is generally greater than that of the vaporization curve at the same state. Rationalize this observation. Note that...
-
Rationalize the following approximate expressions for solid/liquid saturation pressures: (a) P sat sl = A + BT ; (b) P sat sl = A + BlnT
-
(a) A pure substance is described by an expression for G(T, P). Show how to determine Z, U, and C V , in relation to G, T, and P and/or derivatives of G with respect to T and P. (b) A pure substance...
-
QUESTION 3 A business owns seven flats rented out to staff at R500 per month. All flats were tenanted Ist january 21 months rent was in arrears and as at 31st December 14 months' rent wa Identify the...
-
1. 2. 3. Select the Tables sheet, select cells A6:B10, and create range names using the Create from Selection button [Formulas tab, Defined Names group]. Select cells B1:F2 and click the Name box....
-
Tropical Rainwear issues 3,000 shares of its $18 par value preferred stock for cash at $20 per share. Record the issuance of the preferred shares. (If no entry is required for a particular...

Study smarter with the SolutionInn App


