Consider the decomposition reaction of ozone into oxygen: 2O 3 (g) 3O 2 (g) Suppose the
Question:
Consider the decomposition reaction of ozone into oxygen: 2O3(g) → 3O2(g) Suppose the mechanism for this reaction is just the collision between two ozone molecules, as shown in the following elementary step:
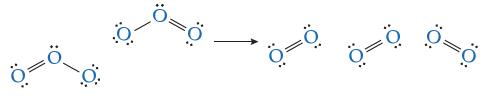
Shown here is a possible transition state. Indicate which bond(s) are breaking and which bond(s) are forming (some lone pairs of electrons are omitted to make the diagram clearer).
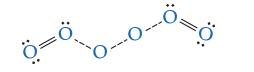
Transcribed Image Text:
11 :O: T :O: ³0: || O: :O:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)

Answered By

Saikumar Ramagiri
Financial accounting:- Journal and ledgers, preparation of trail balance and adjusted trail balance Preparation of income statement, retained earning statement and balance sheet Banks reconciliation statements Financial statement analysis Cash flow statement analysis (both direct and indirect methods) All methods of Depreciations Management Accounting:- Ratios Budgeting control Cash budget and production budget Working capital management Receivable management Costing:- Standard and variance costing Marginal costing and decision making Cost-volume-profit analysis Inventory management (LIFO, FIFO) Preparation and estimation of cost sheet Portfolio management:- Calculation of portfolio standard deviation or risk Calculation of portfolio expected returns CAPM, Beta Financial management:- Time value of money Capital budgeting Cost of capital Leverage analysis and capital structure policies Dividend policy Bond value calculations like YTM, current yield etc International finance:- Derivatives Futures and options Swaps and forwards Business problems Finance problems Education (mention all your degrees, year awarded, Institute/University, field(s) of major): Education Qualification Board/Institution/ University Month/Year of Passing % Secured OPTIONALS/ Major ICWAI(inter) ICWAI inter Pursuing Pursuing - M.com(Finance) Osmania University June 2007 65 Finance & Taxation M B A (Finance) Osmania University Dec 2004 66 Finance & Marketing. B.Com Osmania University June 2002 72 Income Tax, Cost & Mgt, Accountancy, Auditing. Intermediate (XII) Board of Intermediate May 1999 58 Mathematics, Accountancy, Economics. S S C (X) S S C Board. May 1997 74 Mathematics, Social Studies, Science. Tutoring experience: • 10 year experience in online trouble shooting problems related to finance/accountancy. • Since 6 Years working with solution inn as a tutor, I have solved thousands of questions, quick and accuracy Skills (optional): Technical Exposure: MS Office, SQL, Tally, Wings, Focus, Programming with C Financial : Portfolio/Financial Management, Ratio Analysis, Capital Budgeting Stock Valuation & Dividend Policy, Bond Valuations Individual Skills : Proactive Nature, Self Motivative, Clear thought process, Quick problem solving skills, flexible to complex situations. Achievements : 1. I have received an Award certificate from Local Area MLA for the cause of getting 100% marks in Accountancy during my Graduation. 2. I have received a GOLD MEDAL/Scholarship from Home Minister in my MBA for being the “Top Rank student “ of management institute. 3. I received numerous complements and extra pay from various students for trouble shooting their online problems. Other interests/Hobbies (optional): ? Web Surfing ? Sports ? Watching Comics, News channels ? Miniature Collection ? Exploring hidden facts ? Solving riddles and puzzles
4.80+
391+ Reviews
552+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
The gas-phase reaction of NO with F2 to form NOF and F has an activation energy of Ea = 6.3 kJ/mol and a frequency factor of A = 6.0 108 M-1s-1. The reaction is believed to be bimolecular: (a)...
-
The decomposition reaction of N2O5 in carbon tetrachloride is 2 N2O5 -- 4 NO2 + O2. The rate law is first order in N2O5. At the rate constant is 4.82 10-3 s-1. (a) Write the rate law for the...
-
What are the five major realms of China based on climate and geography? How does the natural landscape influence the food and the culture of a nation (using China or a nation familiar to you as an...
-
9. What will the following code print on the console? int main() { } int num = 4; cout < < (num & 3 == 0 ? "first" : "second"); return 0; a. second b. first c. There is a syntax error d. It will...
-
Explain what debit and credit mean.
-
Detecting autocorrelation: von Neumann ratio test.* Assuming that the residual uÌ t are random drawings from normal distribution, von Neumann has shown that for large n, the ratio called the...
-
On December 1,2009, Dresden Company (a U.S. company located in Albany, New York) purchases inventory from a foreign supplier for 60,000 local currency units (LCU). Dresden will pay in 90 days after...
-
Statement of cash flows; investing and financing activities Refer to the situation described in BE 4-13. Prepare the cash flows from investing and financing activities sections of HHC's statement of...
-
According to Hofstede, a feminist culture would b characterized by aggressive accounting treatments. ( Is this statement true or false) course : International Accounting
-
Consider the basic hydrolysis (reaction with aqueous base) of (CH3) 3 CBr. The rate law is first order with respect to (CH 3 ) 3 CBr and zero order with respect to OH . What does this imply about...
-
Explain why the value of k gets larger as the temperature of a reaction mixture is increased.
-
A protective three-layer filter is proposed between the foundation and rock drain located near the toe of a compacted earth-fill dam. Is this filter acceptable? Dis (mm) Dgs (mm) Foundation, finest...
-
Problem Statement | Nessie, the wonder-dog, is sprinting at a constant 10.0 m/s in a straight Useful Equations line. After passing point A, she slows to a stop with a constant acceleration of 1.79...
-
1) How much work does a supermarket checkout attendant do on a can of soup he pushes 0.810 m horizontally with a force of 5.60 N? Express your answer in joules and kilocalories. (For each answer,...
-
You are working as a junior developer at Smashing Websites Ltd. You are part of a team of junior developers that have recently joined the organisation. Smashing Websites provide services to a range...
-
3. Consider the system x + y + bz x+by+4z = 2 = 1 ax+ay + 2z == a In each case, determine all values of a and b which give the indicated number of solutions, if possible. If no such a and b exist,...
-
Income statements for Fanning Company for Year 3 and Year 4 follow. FANNING COMPANY Income Statements Year 4. Sales $200,200 Year 3 $180,200 Cost of goods sold. 143,800 121,800 Selling expenses...
-
Generate a matrix A by setting B = [-1, -1; 1, 1], A = [zeros(2), eye(2); eye(2), B] (a) The matrix A should have eigenvalues 1 = 1 and 2 = -1. Use MATLAB to verify this by computing the reduced row...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Two balls of the same diameter are dropped simultaneously from a very tall bridge. One ball is solid lead, and the other is hollow plastic and has a much smaller mass than the solid lead ball. Use a...
-
Consider a sprinter who starts at rest, accelerates to a maximum speed v max , and then slows to a stop after crossing the finish line. Draw qualitative plots of the acceleration, velocity, and...
-
The bacterium in Figure 3.30 experiences a force due to drag from the surrounding fluid. Does the reaction force to F drag act on (a) The flagellum, (b) The body of the bacterium, or (c) The fluid?...
-
You have just been hired as a new management trainee by Earrings Unlimited, a distributor of earrings to various retail outlets located in shopping malls across the country. In the past, the company...
-
Brief Exercise 10-6 Flint Inc. purchased land, building, and equipment from Laguna Corporation for a cash payment of $327,600. The estimated fair values of the assets are land $62,400, building...
-
"faithful respresentation" is the overriding principle that should be followed in ones prepaparation of IFRS-based financial statement. what is it? explain it fully quoting IAS. how this this...

Study smarter with the SolutionInn App


