(a) Show the orbitals involved in the following thermal electrocyclic reaction. (b) Do the groups rotate in...
Question:
(a) Show the orbitals involved in the following thermal electrocyclic reaction.
(b) Do the groups rotate in a conrotatory or disrotatory manner?
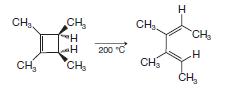
Transcribed Image Text:
CH₂ CH₂ CH₂ TH H CH₂ 200 °C CH₂ CH₂ H CH₂ H CH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a b This is a therma...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Draw and show the overlap of the orbitals involved in the bonds discussed in Problem 36.
-
Show both conrotatory processes for the thermal electtrocyclec conversion of (2E, 4E) - 2,4 -hexadiene into 3,4- dimethylcyclobutene (Eq. 27.8 ). Explain why the two processes are equally likely.
-
The following illustration shows the orbitals used to form the bonds in carbon dioxide. Each color represents a different orbital. Label each or-bital, draw the Lewis structure for carbon dioxide,...
-
Sita lived in a village and could, not afford refrigerator in her house. She knew how to keep water cold and preserve all perishable items in her house. She kept ivet cloth surrounding the earthen...
-
Some ordered pairs are listed in Table 46. Table 46 Some Ordered Pairs Xy 113 3.9 5.8 7.4 9.2 a. Construct a scatterplot by hand. b. Is there a linear association, a nonlinear association, or no...
-
Compute FIFO, LIFO, and Average Cost some of the information found on a detail inventory card for Slatkin Inc. for the first month of operations is as follows. (a) From these data compute the ending...
-
Stanford Corporation began the year with 150,000 shares of common stock outstanding. On March 1, an additional 10,000 shares of common stock were issued. On August 1, another 16,000 shares of common...
-
The bookkeeper for Abduli Company asks you to prepare the following accrued adjusting entries at December 31. 1. Interest on notes payable of $400 is accrued. 2. Services performed but not recorded...
-
The Lo Sun Corporation offers a 6 per cent bond with a current market price ofR875,05. The yield to maturity is 7,34 per cent. The face value is R1 000. Interest is paid semi-annually. How many years...
-
Jimmy owns a garden in which he has planted N trees in a row. After a few years, the trees have grown up and now they have different heights. Jimmy pays much attention to the aesthetics of his...
-
Can you suggest a method for carrying out a stereospecific conversion of trans, trans-2,4-hexadiene into cis,trans-2,4-hexadiene?
-
(a) Write structural formulas for portions of the chain of the atactic, syndiotactic, and iso-tactic forms of polystyrene. (b) If solutions were made of each of these forms of polystyrene, which...
-
The accounts of Apex Consulting, Inc., follow with their normal balances at January 31, 2012. The accounts are listed in no particular order. Account ............... Balance Common...
-
If I add 3 0 , 2 6 7 J of energy to an ideal gas inside a balloon, causing the internal energy to increase by 7 8 1 0 J how much work is done on the environment by the gas is a balloon expands
-
Convert temp from a float to a double using static_cast. float temp 98.6; double dubTemp =
-
You're the Privacy Official (PO) for Peekaboo Hospital (KCH) in Wisconsin.As the PO, you're responsible to see if PH has breached any PHI under HIPAA or state law. On 5/17/2020, you received a call...
-
1. In Brown v. Mississippi , the Court held that the confessions obtained by the police were not admissible as evidence against the defendants. On what constitutional ground did the Court make this...
-
Linda's pizza has been approached by a local hotel to sell them 70 pizzas per day (the local hotel had a kitchen fire, and it will take a year to rebuild their kitchen). At current sales of 100 pizza...
-
A six-column table for JKL Company follows. The first two columns contain the unadjusted trial balance for the company as of July 31, 2016. The last two columns contain the adjusted trial balance as...
-
Suppose that the laptop of Prob. 2.16 is placed in an insulating briefcase with a fully charged battery, but it does not go into sleep mode, and the battery discharges as if the laptop were in use....
-
Explain how the peaks at m/z 115, 101, and 73 arise in the mass spectrum of 3-methy-3-heptanol.
-
Suggest a structure for the compound whose mass spectrum is asfollows: 100 107 81 93 188 70 80 110 120 90 100 130 150 160 170 140 180 190 200 Relative abundance
-
The mass spectra of 3-ethyl-2-pentanone and 4-methyl-2-pentanone are as follows. Explain which spectrum goes with which compound, what is the structure of the ion responsible for the peak at m/z 43...
-
We are evaluating a project that costs $874,800, has a nine-year life, and has no salvage value. Assume that depreciation is straight-line to zero over the life of the project. Sales are projected at...
-
please calculation the following questions. 1. Company A's DPS is $3, EPS is $30, If the company has an ROE of 20%, what is the sustainable growth rate? 2. suppose dividends for a particular company...
-
A transport company purchased a truck for $89,000. The company depreciated the truck at the end of each year at the rate of 19% of its current value. What is the value of the truck at the end of the...

Study smarter with the SolutionInn App


