Which of the following carbocations would you expect to rearrange? a. b. CH2 CH3 CH3 e. CH;CHCHCH3
Question:
Which of the following carbocations would you expect to rearrange?
a.
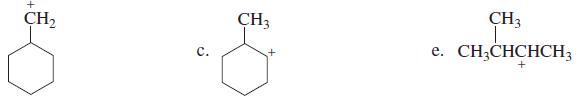
b.

Transcribed Image Text:
CH2 CH3 CH3 e. CH;CHCHCH3 с.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The answer is C After carb...View the full answer

Answered By

Josabanta Jani
I am Josabanta Jani , I completed my Masters in applied geology from NIT Rourkela. During my college time I realised that some of my friends able to teach more perfectly then some of my professors. And some homework and problems were difficult to understand and took lot of time . That time I got inspired to become a tutor , to help students who are scavenger for knowledge.
After college I started teaching in some private classes . And it’s been 2 year doing this tutor job . And I feel like nothing is more interesting then teaching and discussing with other students.
So I truly wish to benefit students . And I response quickly.
Feel free to contact me with questions.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
Which of the following compounds would you expect to be the most generally reactive, and why? DO
-
Which of the following compounds below do you expect to have a longer λ max ?
-
Consider a model of random interest rates R; between year i and i+1. The R, are i.i.d. random variables such that 1+ R; is LogNormal (0.03,0.0010) distributed. Suppose you deposit an amount of 4000...
-
What is the purpose of the reflectron in a time-of-flight mass spectrometer?
-
Understand what is meant by Six Sigma AppendixLO1
-
Suppose you fit a least squares line to 12 data points and the calculated value of SSE is .429. a. Find s2, the estimator of s2 (the variance of the random LO9 error term e). b. Find s, the estimate...
-
If you were trying to assess the financial health of a government administered pension plan, which financial statements or schedules would you review and why?
-
On January 1, 2021, the general ledger of TNT Fireworks includes the following account balances: Credit Debit $ 59,900 27,400 $ 3,400 Accounts Cash Accounts Receivable Allowance for Uncollectible...
-
(Prepared from a situation suggested by Professor John W. Hardy.) Lone Star Meat Packers is a major processor of beef and other meat products. The company has a large amount of T-bone steak on hand,...
-
Tell whether each of the following compounds has the E or the Z configuration: a. b. c. d. H3C CH2CH3 C=C CH3CH2 CH2CH2CI
-
The same alkane is obtained from the catalytic hydrogenation of both alkene A and alkene B. The heat of hydrogenation of alkene A is 29.8 kcal mol (125 kJ mol), and the heat of hydrogenation of...
-
A 0.02% C steel is to be carburized at 1200 C in 4 h, giving 0.45% C at a distance of 0.6 mm beneath the surface. Calculate the carbon content required at the surface of the steel?
-
Q2 2 Points The time between students pinging professor with questions during an exam is modeled by an exponential random variable X (measured in minutes) with parameter (usual notation) Q2.1 1 Point...
-
A. Describe what the formula P = M A E represents. B. What happens if one of these factors becomes deficient? C. In terms of performance, identify the four different types of reinforcement. Provide...
-
1-3.2) K Question 10, 3.1.37 Part 1 of 6 > HW Score: 53.33%, 6.4 of 12 points O Points: 0 of 1 Save Because the mean is very sensitive to extreme values, it is not a resistant measure of center. By...
-
Research the company and obtain the following information: Mission Statement - Purpose of their existence Goals and objectives (What are they in business for) SWOT analysis for this company Based on...
-
please write one page for the concept of organizational structure one page paper of factors affecting organizational structure.
-
Graph the solid bounded by the plane x + y + z = 1 and the paraboloid z = 4 x 2 y 2 and find its exact volume.
-
What can you do to reduce hunger where you live? To reduce hunger globally?
-
Propose a structure for the compound A (C6H15O2 N) that is unstable in aqueous acid and has the following NMR spectra: 2.30 (6H, s): 2.45 (2H. d,J Hz); 3.27 (6H, s): 4.50 (1 H. t. 6 Hz) 6 Proton...
-
Give a curved-arrow mechanism for each of the reamangementreactions.
-
A chemist, Mada Meens, treated ammonia with pentanal in the presence of hydrogen gas and a catalyst in the expectation of obtaining l-pentanamine by reductive amination, In addition to l-pentanamine,...
-
Problem 12.6A (Algo) Liquidation of a partnership LO P5 Kendra, Cogley, and Mel share income and loss in a 3.21 ratio (in ratio form: Kendra, 3/6: Cogley, 2/6; and Mel, 1/6), The partners have...
-
Melody Property Limited owns a right to use land together with a building from 2000 to 2046, and the carrying amount of the property was $5 million with a revaluation surplus of $2 million at the end...
-
Famas Llamas has a weighted average cost of capital of 9.1 percent. The companys cost of equity is 12.6 percent, and its cost of debt is 7.2 percent. The tax rate is 25 percent. What is the companys...

Study smarter with the SolutionInn App


