(S)-Propranolol and (S)-atenolol (Fig. P8.54) are two well-known drugs used as -blockers. They suppress the fight-or-flight response...
Question:
(S)-Propranolol and (S)-atenolol (Fig. P8.54) are two well-known drugs used as β-blockers. They suppress the “fight-or-flight” response and are sometimes used to treat panic in examination or performance situations. Drugs such as these are often characterized by their octanol–water partition coefficients Pow. A small amount of drug is dissolved in a known volume of 1-octanol and then the solution is shaken with a known amount of water. The two phases are allowed to separate, and concentration of drug in each phase is determined. The octanol–water partition coefficient is calculated from the equilibrium concentrations in each phase:
(a) The partition coefficients of these two drugs were determined using water that was made basic to suppress protonation of the amine group. The partition coefficient of one drug is more than 2000 times (3.3 log units) greater than that of the other. Which drug has the greater partition coefficient? Explain.
(b) The partition coefficient of atenolol, expressed in logarithmic units, is log Pow = 0.22. If 1.0 g of atenolol is partitioned between equal volumes of the two phases, how much atenolol is in each phase?
(c) What does your answer to part (a) tell you, if anything, about the relative water solubilities of crystalline atenolol and propranolol? The reported melting points of the two drugs are 152–153°C (atenolol) and 72°C (propranolol).
(d) The amino nitrogen is basic in both drugs. Draw the structure of the conjugate acids of each.
(e) The pKa values of the conjugate acids of both compounds are about the same (9.5). The partition coefficient of each compound was re-determined using a water phase containing a buffer at pH 7.4. How would the partition coefficient of these drugs change (if at all) under these conditions? Would it significantly increase, significantly decrease, or stay about the same? Explain.
(f) These drugs are marketed as their hydrochloride salts (their conjugate acids with a chloride counter-ion). The drugs are administered in the solid phase orally as capsules. What is one reason that the drugs are formulated in this form instead of in their basic form?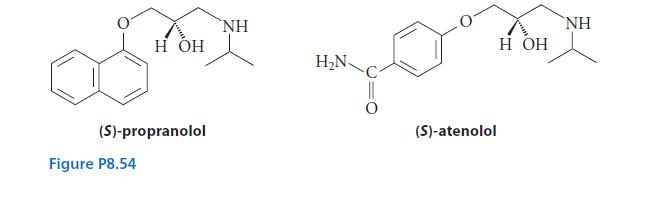
Step by Step Answer:






