Amphotericin B is a powerful antifungal agent used for intravenous treatment of severe fungal infections. Identify the
Question:
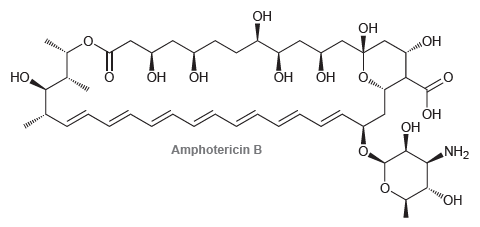
Transcribed Image Text:
ОН ОН Он он ОН ОН ОН Но, ОН Он н Amphotericin B NH2 ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
HO OH ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the most acidic site in thesecompounds: NH2 CH.COCH,CH,CH3 c) a) b) CH3 e) CH;CH,CH,COH d) CH,CH,CCH,CH3
-
Identify the most acidic proton in each of the following compounds: Xo Xx
-
Identify the most acidic hydrogen in each of these compounds: a) HOCCH,CH,SOH 0 CO H 0 I e) CHCCHCOCHCH3 0 b) CH-CH,CH,C=N d) 0 CHOH f) HNCHCOH
-
Show that is one-to-one and find f(x) = J2 1 + 1 dt
-
Locate the annual reports (10-K) of McDonalds Corporation (MCD) for the most recent three years reported (2013, 2014, &2015). For this assignment, do NOT use the Dupont Method (if applicable). Using...
-
Let f (x) = 3 x and g(x) = 1/x 2 . Calculate the following functions. Take x > 0. g( f (x))
-
Reliability. You are laboring through a chemistry laboratory assignment in which you measure the conductivity of a solution. What does it mean for your measurement to be reliable? How can you improve...
-
Direct Manufacturing Labor and Variable Manufacturing Overhead Variances. Sarah Beths Art Supply Company produces various types of paints. Actual direct manufacturing labor hours in the factory that...
-
Swifty Corporation incurred the following transactions. Purchased raw materials on account $ 5 5 , 8 0 0 . Raw Materials of $ 3 7 , 6 0 0 were requisitioned to the factory. An analysis of the...
-
Allie has bought a new apple orchard. The orchard has a single file of trees, numbered from 1 to N. Each tree has a certail number of ripe apples. Allie has a rule she wants to follow. She wants to...
-
The following compound is one of the strongest known acids: (a) Explain why it is such a strong acid. (b) Suggest a modification to the structure that would render the compound even more acidic. II...
-
Predict the position of equilibrium for each of the following reactions: (a) (b) (c) HO NH NH2
-
Calculate the specific activities of Na24 and U235 nuclides whose half-life are 15 hours and 7.1.10 s years respectively.
-
Account is a domestic growth portfolio. The current holdings are primarily US exchange-traded stocks and bonds. To remain in compliance, the total portfolio may only invest up to a maximum of 5% in...
-
The President of the United States needs your help. He has asked you to investigate and find answers to several important questions. His questions are included in the Letter from the President below....
-
Cheng Co. reports the following information for the coming year. Labor rate, including fringe benefits Annual labor hours Annual materials purchases Annual overhead costs: Materials purchasing,...
-
Compared to most objects, sound waves travel very fast. It is fast enough that measuring the speed of sound is a technical challenge. One method you could use would be to time an echo. For example,...
-
Sharif and Judith are married and purchased a vacation home together in Maine for $ 2 5 0 , 0 0 0 . Sharif died suddenly six months later and at that time the fair market value of the vacation home...
-
Solve each system. 0 = N 15/00 + -7 || 1 2- N 1100 1 + - -5 + X = 2. Nim -Tin min -
-
Catalytic hydrogenation of naphthalene over PdC results in rapid addition of 2 moles of H 2 . Propose a structure for this product.
-
Provide the reagents necessary for the following syntheses. More than one step may be required. (a) (b) (c) (d) (e) (f) HO HO Br OH HO HO CI OH
-
Predict the major product from each of the following reactions. (a) (b) (c) (d) (e) (f) OH SOC, pyr OH HBr NaNH2 OH OH (1) TsCI, pyr (2) EtSNa Nal, H2SO OH
-
The following reaction does not produce the product shown. (a) Predict the major product from the conditions shown above, and write a detailed mechanism for its formation. (b) What reaction...
-
Comfort Golf Products is considering whether to upgrade its equipment Managers are considering two options. Equipment manufactured by Stenback Inc. costs $1,000,000 and will last five years and have...
-
Weaver Corporation had the following stock issued and outstanding at January 1, Year 1: 71,000 shares of $10 par common stock. 8,500 shares of $60 par, 6 percent, noncumulative preferred stock. On...
-
Read the following case and then answer questions On 1 January 2016 a company purchased a machine at a cost of $3,000. Its useful life is estimated to be 10 years and then it has a residual value of...

Study smarter with the SolutionInn App


