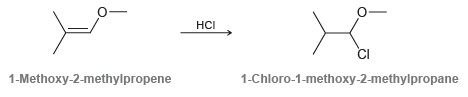
When 1-methoxy-2-methylpropene is treated with HCl, the major product is 1-chloro-1-methoxy-2-methylpropane. Although this reaction proceeds via an
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





