1.10 moles of N 2 at 20.5°C and 6.20 bar undergoes a transformation to the state described...
Question:
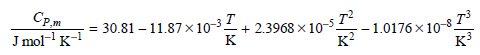
Transcribed Image Text:
Сри Jmol-1 к-1 + 2.3968 x 10- г? -1.0176 х 10-3. 30.81-11.87х10 3. -3T к? кз к
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Cpm Jmol K 3081 118710 23968 x 105 T Pf ASnR In n P T 48815 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The standard entropy of Pb(s) at 298.15 K is 64.80 J K -1 mol - 1 . Assume that the heat capacity of Pb(s) is given by The melting point is 327.4C and the heat of fusion under these conditions is...
-
A 2.5 x 2.5 m steel sheet 1.5 mm thick is removed from an annealing oven at a uniform temperature of 425?C and placed in a large room at 20?C in a horizontal position.(a) Calculate the rate of heat...
-
A plastic tube of 7.6-cm ID and 1.27 cm wall thickness having a thermal conductivity of 1.7 W/(m K), a density of 2400 kg/m3, and a specific heat of 1675 J/(kg K) is cooled from an initial...
-
Mr. Silkwallah established the Fashion Clothing Company (FCC) to market designer clothes. The business was to get designer clothes produced by tailors, exclusively for FCC. FCC provides the following...
-
What behaviors do effective leaders exhibit?
-
Evaluate exactly the given expressions if possible. cot 1 3
-
2 Select your consumer-oriented sales promotion activities.
-
You Can Paint Too prepares and packages paint products. You Can Paint Too has two departments: Blending and Packaging. Direct materials are added at the beginning of the blending process (dyes) and...
-
Please help farmer Lupe calculate the following accrual adjustments in the table (change in value portion). I completed the first example to help you. (2 points; .5 point per highlighted box) Item (+...
-
A continuous random variable X that can assume values between x = 1 and x = 3 has a density function given by f(x) = 1/2. (a) Show that the area under the curve is equal to 1. (b) Find P(2 < X <...
-
What is design capacity? Effective capacity? Best operating level?
-
Propose a plausible synthesis for the following transformation. CH3 CH3 CH3
-
There are 16 possible binary structures on the set {a, b} of two elements. How many nonisomorphic (that is, structurally different) structures are there among these 16? Phrased more precisely in...
-
Briefly explain the difference between a k-factor model and the capital asset pricing model
-
Refer to the cost data, Picture below. Take off the square feet of wall forms and cubic yards of ready mix concrete for the walls of the elevator pit. Determine the total material and labor cost for...
-
possible Submit quiz A researcher studies water clarity at the same location in a lake on the same dates during the course of a year and repeats the measurements on the same dates 5 years later. The...
-
A liquid hydrocarbon mixture was made by adding 295 kg of benzene, 289 kg of toluene and 287 kg of p-xylene. Assume there is no change of volume upon mixing, i.e., Vmix=0 , in order to determine: 1....
-
b) Maseru Development Bank has R850 million credit with Matsieng Hydroelectric Power, with a maturity of eighteen months. The expected loss for Maseru Development Bank is R22 million, and the...
-
Solve each rational inequality in Exercises 4360 and graph the solution set on a real number line. Express each solution set in interval notation. x + 5 x + 2 < 0
-
QUESTION 9 HC-O-C-R R-C-O-CH HC-O-P-O-CH-CH-NH3* O || O a. Phosphatidic acid, Serine O b. Lysophosphatidic acid, Serine, Free FA O c. Lysophosphatidylserine, Free FA O d. 2 Free FAs, Serine, Glycerol...
-
Find the three cube roots of 3.000 2.000i .
-
Find the four fourth roots of 3.000i.
-
Find the real and imaginary parts of (3.00 + i) 3 + (6.00 + 5.00i) 2 . Find z .
-
You have just been hired as a new management trainee by Earrings Unlimited, a distributor of earrings to various retail outlets located in shopping malls across the country. In the past, the company...
-
Brief Exercise 10-6 Flint Inc. purchased land, building, and equipment from Laguna Corporation for a cash payment of $327,600. The estimated fair values of the assets are land $62,400, building...
-
"faithful respresentation" is the overriding principle that should be followed in ones prepaparation of IFRS-based financial statement. what is it? explain it fully quoting IAS. how this this...

Study smarter with the SolutionInn App


