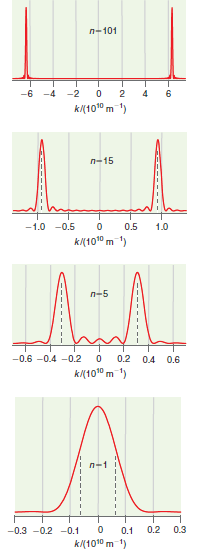
Consider the results of Figure 17.5 more quantitatively. Describe the values of x and k by x
Question:
Figure 17.5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





