The following diagram shows the energies of valence molecular orbitals of boron trifluoride. The energies of three
Question:
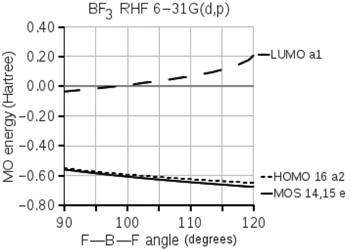
Transcribed Image Text:
BF3 RHF 6-31G(d,p) 0.40 0.20 -LUMO a1 0.00 :-0.20- 5 -0.40 - -0.60 --номо 16 а2 -MOS 14,15 e -0.80 90 100 110 120 F-B-F angle (degrees) MO energy (Hartree)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
BF 3 has 32 electrons which fill up through MOs 14 15 ...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following diagram shows one-half of a restriction site. (a) Draw the other half. (b) Use heavy arrows () to identify type II cleavage sites that would yield blunt-ended duplex DNA products. (c)...
-
Consider the molecular orbitals of the P2 molecule. Assume that the MOs of di-atomics from the third row of the periodic table are analogous to those from the second row. (a) Which valence atomic...
-
The iodine bromide molecule, IBr, is an inter-halogen compound. Assume that the molecular orbitals of IBr are analogous to the homo nuclear diatomic molecule F2. (a) Which valence atomic orbitals of...
-
Gems Co. uses the indirect method to prepare its statement of cash flows. The following comparative statement of financial position for 2021 and 2022 are presented: At December 31 2022 2021 Property,...
-
Describe the error. cos 60o = opp / hyp = 1 / 2
-
The stockholders' equity section of Lachlin Corporation's balance sheet at December 31 is presented here. Instructions From a review of the stockholders' equity section, answer the following...
-
In an automated payroll processing environment, a department manager substituted the time card for a terminated employee with a time card for a fictitious employee. The fictitious employee had the...
-
Springfield mogul Montgomery Burns, age 80, wants to retire at age 100 so he can steal candy from babies full time. Once Mr. Burns retires, he wants to withdraw $1 billion at the beginning of each...
-
Find the size of the permanent endowment needed to generate an annual $25,000 forever at a continuous interest rate of 5%. $
-
A. State the principle of conservation of momentum and state the condition under which it is valid. B. An arrow of mass 0.25 kg is fired horizontally towards an apple of mass 0.10 kg that is hanging...
-
The energy of the occupied valence MOs of H 2 S is shown as a function of the HSH bond angle. Compared to the analogous diagram, Figure 24.11, for H 2 O, the 2a1 MO energy decreases more as the bond...
-
The density of states (DOS) of pyrite, crystalline FeS 2 (as calculated by Eyert et al, Physical Review B 55 (1998): 6350, is shown next. The highest occupied energy level corresponds to zero energy....
-
The Hall effect experimental arrangement was described in Example 11.4. The required diff-amp is to be designed in the circuit configuration in Figure P11.35. The transistor parameters are \(V_{T...
-
You're shopping for a new car. One car that you really liked costs $28,000. You are planning to fully finance this purchase. The loan contract requires fixed monthly payments for 4 years, at 5...
-
(4c) Assume equidistant meshes and reconstruction of slopes by a particular av- eraging. More precisely, the M-piecewise cubic function s is to satisfy the generalized interpolation conditions s(xj)...
-
Victoria goes into Elise's computer store Byte-Me Computers to have her computer's RAM upgraded. She tells Elise she needs the computer by the end of the day to work on a BSB151 assignment due the...
-
Search and download datasets from the internet by topic Retail " ( for example: kaggle datasets and so on ) . Then determine what problems can be raised based on the dataset. 2 . Carry out the Big...
-
S Based on MD5 hash function, if the input is "Under the PRISM program, which started NSA gathers Internet communications from foreign targets from nine major U.S. Internet-based communication ervice...
-
In 1935, J. Bredt, a German chemist, proposed that a bicycloalkene cannot have a double bond at a bridgehead carbon unless one of the rings contains at least eight carbon atoms. This is known as...
-
A regular deposit of $100 is made at the beginning of each year for 20 years. Simple interest is calculated at i% per year for the 20 years. At the end of the 20-year period, the total interest in...
-
The mobilities of H + and Cl at 25C in water are 3.623 10 7 m 2 s 1 V 1 and 7.91 10 8 m 2 s 1 V 1 , respectively. What proportion of the current is carried by the protons in 10 3 m HCl(aq)? What...
-
A space vehicle of internal volume 3.0 m 3 is struck by a meteor and a hole of radius 0.10 mm is formed. If the oxygen pressure within the vehicle is initially 80 kPa and its temperature 298 K, how...
-
A manometer was connected to a bulb containing carbon dioxide under slight pressure. The gas was allowed to escape through a small pinhole, and the time for the manometer reading to drop from 75 cm...
-
Which of the following groups should be the focal point of a company's strategy? MulABC Inc. produces a single product, In doing so, the company incurs overhead costs as follows: Indirect Factory...
-
When capturing and documenting system audit requirements for an IT project, the teams is focused on capturing the following
-
Gabriela is a claims adjuster who is conducting a phone interview with a claimant. She has asked a question to openly address an issue and clarify one of the claimant's statements. Gabriella has...

Study smarter with the SolutionInn App


