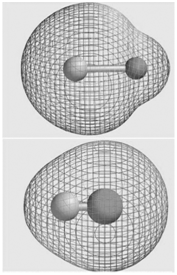
Question: The molecular electrostatic potential maps for LiH and HF are shown here. Does the apparent size of the hydrogen atom (shown as a white sphere)
The molecular electrostatic potential maps for LiH and HF are shown here. Does the apparent size of the hydrogen atom (shown as a white sphere) tell you whether it is an electron acceptor or an electron donor in these molecules?
Step by Step Solution
3.37 Rating (166 Votes )
There are 3 Steps involved in it
Yes it does The electrostatic potential is displayed on a tot... View full answer

Get step-by-step solutions from verified subject matter experts


